All Of The Following Are Characteristics Of Igm Except

Ever feel like your immune system is putting on a grand, sometimes chaotic, opera? Well, you’re not alone! Our bodies are constantly orchestrating a symphony of defense, and one of its earliest and most energetic performers is the mighty Immunoglobulin M, or IgM for short. Think of it as the opening act, the one that rushes onto the stage with a big, bold statement, ready to tackle whatever comes its way.
But like any star player, IgM has its quirks and its defining moments. And sometimes, in the whirlwind of scientific discovery, we learn what’s truly part of its performance and what’s just a bit of stage dressing. So, let’s dive into the fascinating world of IgM, and figure out which of its supposed characteristics are a total hit, and which ones… well, let’s just say they might be a swing and a miss.
The Grand Entrance: IgM’s Role as the First Responder
Imagine you’ve just discovered a new café in your neighborhood. The very first time you walk in, the staff are extra attentive, right? They want to make sure you have a great experience and remember them. That’s pretty much IgM’s vibe when your body encounters a new invader, like a pesky virus or a rogue bacterium.
IgM is your immune system’s first responder. It’s the earliest antibody produced during an infection. When your body flags an unfamiliar antigen (think of antigens as the unique "uniforms" worn by germs), IgM is the first type of antibody to be synthesized by your B cells. It's like the welcome wagon, arriving with a flourish to say, "Hey, we see you, and we're on it!"
This swift response is crucial. It buys your immune system valuable time to ramp up production of other, more specialized antibodies, and to mount a more targeted defense. Without IgM’s quick arrival, those pathogens could really get a head start.
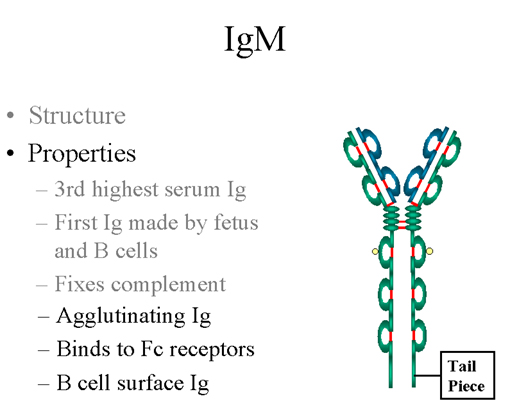
The Shape of Things: IgM’s Unique Structure
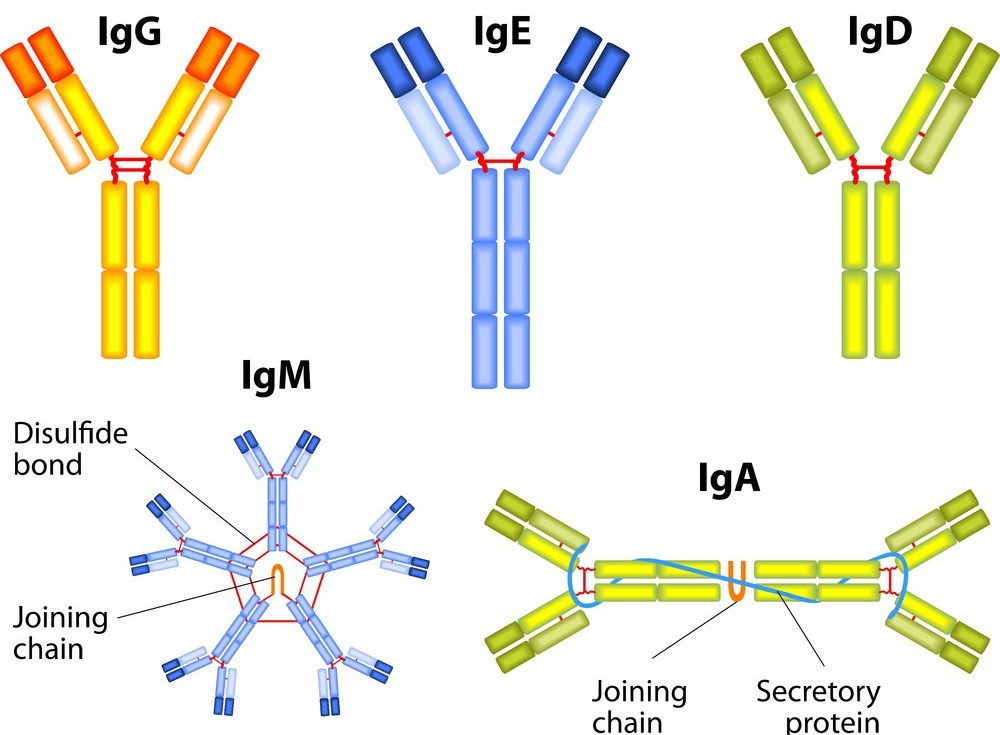
One of the most striking things about IgM is its shape. Unlike most other antibodies, which are Y-shaped (monomers), IgM typically exists as a pentamer. What's a pentamer, you ask? Well, it’s like five of those Y-shaped antibodies joined together in a ring, often with a little extra piece called a J chain holding them all in place. Picture a five-pointed star, or maybe a really fancy chandelier.
This unique, multi-armed structure is a significant advantage. Each of those arms can bind to an antigen. So, a single IgM molecule has the potential to bind to five different sites on invading pathogens simultaneously. This multivalency makes IgM incredibly effective at agglutination – that’s a fancy word for clumping things together. Think of it like a giant net, scooping up and immobilizing multiple germs at once.
This clumping action is a big deal. It makes it easier for other parts of the immune system, like phagocytes (the "clean-up crew" cells), to find and engulf the pathogens. It's like putting a giant flashing neon sign on the invaders, saying "Easy target here!"

The Power of the Pentamer: Complement Activation
Beyond just clumping, IgM is also a powerhouse when it comes to activating the complement system. The complement system is another crucial arm of your innate and adaptive immunity, composed of a cascade of proteins that work together to neutralize or eliminate pathogens.
When IgM binds to a pathogen, it can initiate a chain reaction that leads to the activation of these complement proteins. This can result in several things: the pathogen can be directly lysed (burst open), it can be marked for destruction by phagocytes (opsonization), or it can trigger inflammation to recruit more immune cells to the site. It’s like IgM is the conductor of an orchestra, signaling all the different instruments in the complement system to play their part.
This ability to efficiently activate complement is a hallmark of IgM and makes it incredibly potent in fighting off bacterial infections, especially those with encapsulated bacteria. It's a sophisticated, multi-layered defense strategy, and IgM is at the forefront of it.
IgM’s Affinity: A Love-Hate Relationship
Now, here's where things get a little nuanced. When it comes to affinity – which is essentially how tightly an antibody binds to its antigen – IgM is often described as having a lower affinity per individual binding site compared to antibodies like IgG. However, because it has so many binding sites (remember the pentamer?), its avidity (the overall strength of binding) is very high.
Think of it like this: a single Lego brick might not hold together super strongly, but a Lego structure with many bricks locked in place is incredibly robust. IgM, with its multiple binding points, creates a very strong overall connection to the pathogen, even if each individual "lock" isn't the tightest.
This might seem counterintuitive, but it’s a clever evolutionary trick. High affinity isn't always needed for the initial capture and flagging of a pathogen. The rapid generation and broad binding power of IgM are more important for that first wave. As the immune response matures, B cells can undergo a process called affinity maturation, which leads to the production of higher-affinity IgG antibodies.
The Lifetime of an Antibody: IgM’s Duration
So, how long does IgM stick around? This is another key characteristic. IgM is a transient antibody. Its presence in the blood indicates a recent or current infection. Once the acute phase of an infection passes and the B cells switch to producing other antibody types like IgG, the levels of IgM typically decline.
This is why IgM levels are so important in diagnostic tests. Detecting IgM can tell doctors that an infection is new, while the presence of IgG might indicate a past infection or a successful vaccination. It’s like a timestamp on your immune response, giving valuable clues about the history of your body’s battles.
The IgG Imposter? Let’s Talk Exceptions.
Alright, we've laid out the impressive resume of IgM. Now, let's get to the nitty-gritty. In the study of immunology, it’s not uncommon to encounter statements about antibodies, and sometimes, those statements are designed to test your understanding of their specific roles and characteristics. So, let's consider what would be an exception to the typical profile of IgM.
If we're talking about characteristics that are not typically associated with IgM, what might come to mind? Well, let’s consider a few possibilities:

1. Crosses the Placenta
This is a big one. One of the most well-known and vital functions of IgG antibodies is their ability to cross the maternal placenta. This transfer of IgG from mother to fetus provides crucial passive immunity, protecting the newborn in the critical early months of life. Think of it as a lifelong inheritance of protection.
IgM, on the other hand, does not readily cross the placenta. This is an important distinction. If a newborn has detectable IgM against a specific pathogen, it often indicates that the fetus itself has been infected in utero, rather than simply receiving maternal antibodies. This is a critical diagnostic clue for congenital infections.
So, if you see a statement claiming that IgM crosses the placenta, that’s a definite “Except!” It’s a fundamental difference between IgM and IgG, and a key diagnostic marker.
2. Long-Term Immunity
We’ve already touched on this. IgM is your "hello, nice to meet you, let's get this party started!" antibody. It’s there for the initial rush. Long-term immunity, the kind that protects you for years after an infection or vaccination, is primarily mediated by memory B cells and the production of long-lived IgG (and sometimes IgA) antibodies.
While IgM plays a vital role in clearing the initial infection, it doesn’t hang around to provide that lasting shield. If a statement suggested that IgM is the primary antibody responsible for long-term immunological memory or sustained protection, that would be an exception. It’s the early bird, not the long-haul traveler.
3. High Affinity per Binding Site
As we discussed, while IgM has high avidity due to its multiple binding sites, the affinity of each individual binding site is generally lower than that of mature IgG antibodies. The immune system specifically upregulates affinity through somatic hypermutation and affinity maturation as the response progresses. Therefore, a characteristic stating that IgM possesses very high affinity per binding site would also be an exception. It’s the collective power, not the individual strength, that’s IgM's initial game.
4. Primary Mediator of Opsonization for Long-Term Memory Cells
While IgM can contribute to opsonization (marking pathogens for phagocytosis), it's not its primary, long-term role in the context of immunological memory. Opsonization for sustained protection and memory recall is more strongly associated with IgG, which is produced in higher quantities and has higher affinity. So, if you were presented with a characteristic that positioned IgM as the primary mediator of opsonization for long-term memory, that would be another exception. IgM does its job powerfully, but IgG often takes the baton for the marathon of ongoing immunity.
Putting It All Together: A Daily Dose of Immunology
So, there you have it. IgM is the enthusiastic, five-armed wonder of the immune system, kicking off the party when a new threat appears. It clumps, it calls for backup, and it announces its presence with a quick, energetic burst.
The key takeaway is to remember that while IgM is a fantastic first responder, certain functions like placental transfer, long-term immunity, and particularly high affinity per binding site are the domain of other immunoglobulins, most notably IgG. These distinctions aren't just trivia; they’re the very foundations of how our immune system works and how we diagnose and treat diseases.
Think about it in your daily life. When you feel that first tickle of a cold coming on, it’s your IgM saying, "Here we go!" When you're later feeling better and the risk has passed, it's the IgG and memory cells keeping you safe from round two. It’s a beautiful, complex dance, and every player has its starring role.
Understanding these nuances reminds us of the incredible sophistication of our own biology. It’s like learning the difference between a vibrant opening number and a powerful, enduring ballad. Both are essential to the performance, but they serve different purposes, at different times, and with different strengths. And that, in its own way, is pretty amazing to ponder over your morning coffee.
