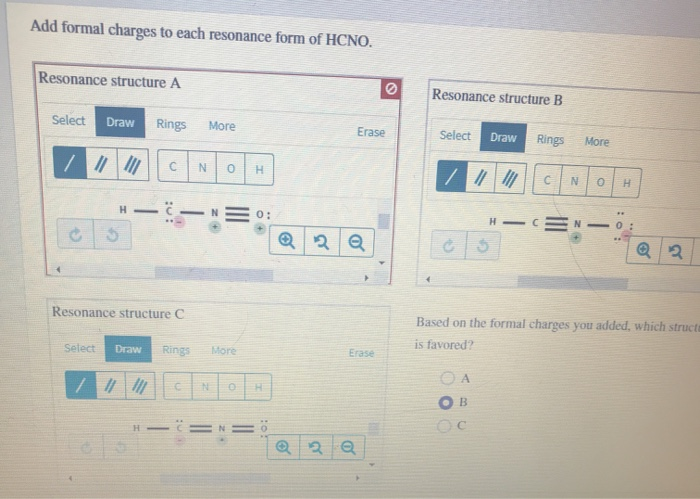
Add Formal Charges To Each Resonance Form Of Hcno.

Hey there, science curious folks! Today, we're diving into something that might sound a little fancy, but trust me, it's actually a whole lot of fun. We're going to talk about HCNO and its cool little trick called resonance. Think of it like a molecule playing dress-up, showing off different outfits while still being the same old molecule underneath. Pretty neat, right?
Now, you might be wondering, "What in the world is HCNO?" Well, it's a tiny little guy, a molecule made of hydrogen (H), carbon (C), nitrogen (N), and oxygen (O). Simple enough! But when these atoms get together, they can arrange themselves in a few different ways. It’s like having building blocks and being able to connect them in slightly different patterns. This is where the magic of resonance comes in.
Imagine you have a favorite toy. Sometimes you might hold it this way, and other times that way, but it's still your favorite toy, right? Molecules can be a bit like that. For HCNO, there are a few ways its electrons can be arranged, and these different arrangements are called resonance structures or resonance forms. It's not that the molecule is actually switching between these forms back and forth super fast. Instead, it’s like a blend, a happy medium where the real molecule is somewhere in between all these different pictures we draw.
So, what makes this so entertaining? It’s all about how we chemists like to represent these invisible things. When we draw molecules, we often use dots and lines to show where the electrons are hanging out. For HCNO, a single way of drawing it doesn't quite tell the whole story. It’s like trying to describe a vibrant painting by only showing one corner of it. You're missing the bigger, more colorful picture!
This is where we get to add something called formal charges. Don't let the name scare you! Think of formal charges like little “thanks” or “oops” labels we put on the atoms in our drawings. They help us understand how the electrons are distributed and which atoms are feeling a bit more "positive" or "negative" in that particular drawing. It's a way of keeping score, if you will, to make sure our drawings are balanced and make sense.
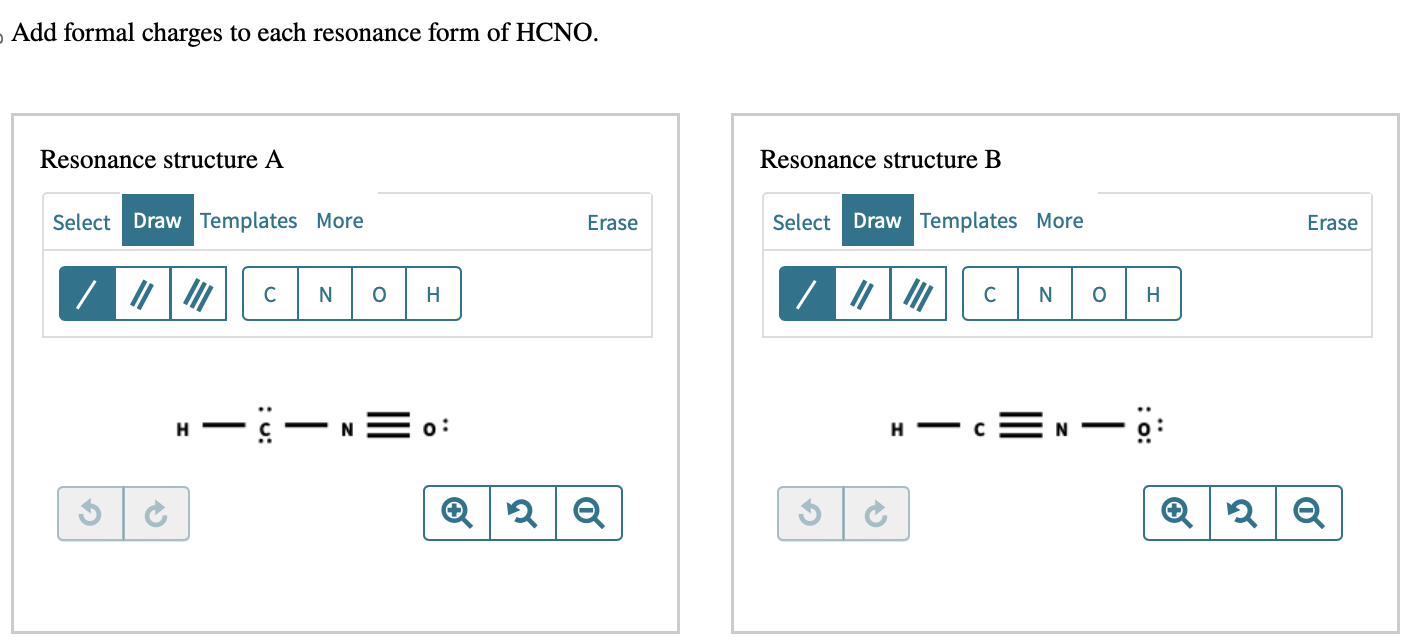
When we draw the different resonance forms of HCNO, we can see that some atoms end up with these little formal charge labels. Some might have a little positive sign (+), meaning they're a bit electron-deficient in that picture. Others might have a little negative sign (-), indicating they're hogging a few extra electrons in that particular arrangement. And some atoms, lucky ducks, might have a zero charge, meaning they're perfectly balanced in that drawing.
Let's break down why this is so cool. Imagine you have three friends, and each friend is holding a different toy. You can draw each friend with their toy, right? Now, what if your friends could magically swap their toys around? That’s kind of like what happens with electrons in resonance. The electrons are shared and moved around, creating these different possibilities. By adding the formal charges to each of these possibilities, we get a clearer picture of where the "action" is happening in the molecule.

It's like solving a puzzle. You have these different pieces, these different drawings of HCNO. And by putting the formal charges on them, you start to see how they fit together. You realize that one drawing might have a positive charge on the carbon, and another might have a negative charge on the oxygen. It helps you understand the molecule’s personality, its tendencies, and how it might interact with other molecules. It’s a peek behind the curtain of chemical behavior!
Think of it as giving each drawing a little score card. This score card tells us which atoms are happy campers (neutral charge) and which ones are feeling a bit more dramatic (carrying a positive or negative charge). It's a really insightful way to look at the same molecule from different angles!
(Solved) - Add formal charges to each resonance form of HCNO. Resonance
The fun part is that no single drawing perfectly represents HCNO. The real molecule is a sort of average of all these resonance forms, a blend that’s more stable and happy than any single drawing alone. So, when we add those formal charges to each resonance form, we're not just doing a bookkeeping exercise. We're actually learning about the true nature of HCNO, its electron distribution, and why it behaves the way it does.
This might seem a bit abstract, but it has real-world implications. Understanding these electron arrangements and charges helps scientists design new materials, develop medicines, and even figure out how chemical reactions happen. So, the next time you hear about resonance and formal charges, don't think of it as dry chemistry. Think of it as a fun, visual puzzle that helps us understand the amazing and complex world of molecules like HCNO!
It’s a little bit of molecular detective work, adding clues (the formal charges) to our suspects (the resonance forms) to uncover the truth about this fascinating little molecule. Give it a look, and you might find yourself as captivated as we are!