Acids Bases And Salts From The Elements Of Chemistry Series
Hey there, science curious folks! Ever feel like chemistry is this big, scary, lab-coat-wearing thing that’s way beyond your reach? Well, get ready to have your mind tickled, because we're about to dive into the wonderfully wacky world of acids, bases, and salts. And guess what? It’s not just for mad scientists in white coats – it’s actually all around you, making your life, dare I say, way more interesting!
Think about it. That tangy lemonade you sip on a hot day? Yep, that’s an acid at work. Your super-sudsy dish soap? Hello, base! And that little pinch of salt you add to your popcorn? You guessed it – a classic salt. See? You’re already a chemistry whiz without even realizing it! This is all part of the fantastic "Elements of Chemistry" series, and trust me, these building blocks of our world are anything but boring.
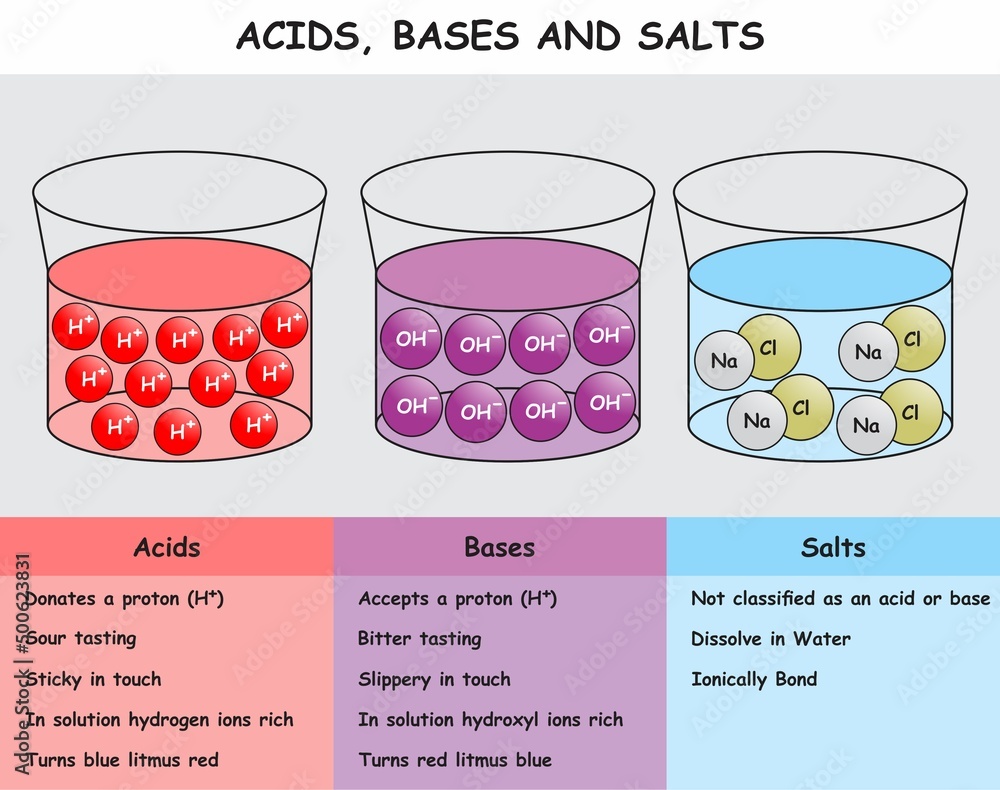
So, what’s the deal with acids? Imagine tiny little hydrogen ions (H+) just itching to get out and play. When something has a lot of these eager little guys, we call it an acid. They tend to have a sour taste (think lemons, vinegar – but please, don’t go tasting random chemicals, okay?). Acids are also super good at dissolving things. That’s why acids are used in everything from cleaning toilets (don't worry, they're diluted!) to helping digest your food.
But don't get too bogged down in the "sour" aspect! Acids are also the secret ingredient in many of your favorite treats. That fizz in your soda? Often thanks to carbonic acid. That delightful tang in yogurt? Lactic acid, of course! They’re like the little zing that makes things pop. We’re talking about things like citric acid in oranges and limes, acetic acid in vinegar, and hydrochloric acid in your stomach (which is incredibly important for breaking down food – go, science!).
Now, let’s swing over to the world of bases. If acids are all about giving away those H+ ions, bases are the opposite – they’re either ready to accept H+ ions or they have these things called hydroxide ions (OH-) that are also ready for action. Bases tend to feel slippery (ever noticed how soap feels that way? Bingo!) and often have a bitter taste (again, don’t go around tasting things!).

Bases are the unsung heroes of cleanliness and comfort. They’re brilliant at neutralizing things, which is a super important concept we’ll get to in a sec. Think about that baking soda paste you might use to clean your oven – that's a base! Or even that antacid you might take for heartburn? That’s your body getting a little help from a base to calm down an overactive acid. Amazing, right?
So, what happens when these two opposing forces, acids and bases, meet? It’s like a friendly chemical handshake, a beautiful dance of molecules! This is where the magic of neutralization happens. When an acid and a base react, they cancel each other out, forming something completely new and generally quite pleasant: salts and water.

And when we say "salts," don't just think of the white stuff on your fries! Salts are actually a whole huge family of compounds formed from the reaction of an acid and a base. They can be anything from the everyday table salt (sodium chloride, a reaction between hydrochloric acid and sodium hydroxide – fancy!) to more complex compounds that are crucial for everything from our bodies to industrial processes.
Think about the minerals in your body – many of them are actually salts! They're essential for nerve function, muscle contraction, and keeping your bones strong. So, that pinch of salt you add to your food isn't just for flavor; it's also a tiny contribution to keeping you healthy and happy. How cool is that?

The "Elements of Chemistry" series really shines here because it breaks down these concepts so clearly. It shows you how these seemingly simple reactions are the bedrock of so much of what we experience. It’s not just abstract theory; it’s about understanding the world around you on a deeper, more engaging level.
Why is this so inspiring? Because understanding acids, bases, and salts unlocks a new way of seeing. You start to appreciate the chemical wonders in your kitchen, the cleaning power of everyday substances, and even the fundamental processes that keep you alive. It’s like gaining a secret superpower – the power of knowledge!
.png)
Imagine the fun you can have! You can start to understand why certain cleaning products work, why your garden soil might need adjusting, or even why some foods taste the way they do. It’s about empowering yourself with information, and that, my friends, is always an inspiring journey. The more you learn about these fundamental chemical building blocks, the more you realize how interconnected everything is.
So, next time you’re enjoying a sour candy, using some baking soda to clean, or even just adding salt to your meal, take a moment. Give a little nod to the amazing world of acids, bases, and salts. They’re the unsung heroes, the quiet architects of our everyday lives, and understanding them is like opening a door to a whole new universe of understanding and wonder.
Don't be intimidated by chemistry. Embrace it! Dive into the "Elements of Chemistry" series, explore these fascinating topics further. You might just discover a passion you never knew you had. The world is a chemical wonderland, and you've got the key to unlocking its secrets. Go forth, be curious, and let the learning ignite your spark!
