Acid-base Precipitation And Redox Reactions Worksheet
Ever feel like your kitchen sink is staging a dramatic showdown? You know, one minute it's all bubbly and singing the praises of dish soap, and the next, it's like a grumpy old man muttering about clogs and mysterious goo. Well, buckle up, buttercup, because we're about to dive into the wild and wacky world of acid-base precipitation and redox reactions, and trust me, it's more relatable than you think!
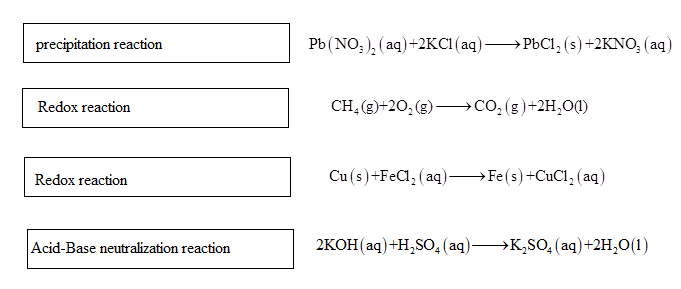
Think of your sink as a mini-chemical laboratory. When you pour that super-duper acidic drain cleaner down, it’s like unleashing a tiny, angry scientist determined to dissolve anything in its path. That’s your acid, ready to donate protons like they’re free samples at Costco. On the other side, you’ve got your bases – think baking soda, or even that slightly slippery residue left from, say, a particularly enthusiastic spaghetti sauce incident. Bases are the chill ones, the ones that are happy to accept those protons. When they meet? Neutralization station! It’s like a friendly handshake between opposites, forming something much more mellow, usually water and a salt. Remember when you were a kid and mixed baking soda and vinegar? That fizzy explosion? That’s a classic, albeit slightly chaotic, acid-base reaction in action. Your parents probably loved that, right? (Wink, wink).
Now, precipitation. This is where things get a little… clumpy. Imagine you’re making a fancy latte, and you accidentally pour in some milk that's gone a tad off. What happens? You don't get a smooth, creamy beverage, do you? You get… well, let's just say it's not Instagram-worthy. Those little curds are basically precipitates. In the world of chemistry, precipitation is when two soluble things get together, decide they’d rather not be dissolved anymore, and form a solid. It’s like they’re throwing a mini-party for themselves, kicking out all the freeloading water molecules. So, when you're looking at a worksheet about it, picture those uninvited guests at a party, forming their own little clique at the bottom of the beaker.
Why is this important? Well, think about those stubborn mineral deposits in your shower. That chalky white stuff? That's often a precipitate formed from dissolved minerals in the water reacting with other things. It’s like the universe decided to decorate your bathroom with tiny, unwanted sculptures. Cleaning it involves either dissolving it with an acid (more sink drama!) or sometimes physically scrubbing it away. It's a constant battle between solubility and insolubility, a microscopic turf war happening right before your eyes.
The Zany Dance of Electron Swapping
Alright, let’s switch gears and talk about redox reactions. These are the rock stars of the chemistry world, all about the thrilling exchange of electrons. Think of electrons as the popular kids at school, constantly being passed around, causing all sorts of commotion. In a redox reaction, one substance loses electrons (it gets oxidized, which sounds bad, but it's like the athlete who gives it their all and gets a medal for effort) and another substance *gains electrons (it gets reduced, which sounds like a downer, but it's actually a good thing, like the one who catches the winning pass).
The simplest way to remember this is the good old mnemonic: OIL RIG. Oxidation Is Loss (of electrons), and Reduction Is Gain (of electrons). It’s like a cosmic game of hot potato, but with tiny, energetic particles. Who’s giving the hot potato? The oxidizing agent. Who’s bravely grabbing it? The reducing agent.

Ever seen a rusty old bicycle? That’s oxidation in action! The iron in the metal is losing electrons to oxygen in the air, forming that lovely reddish-brown flaky stuff. It’s like the bicycle is slowly giving up its metallic sheen, transforming into something… well, less shiny. And that’s just one example. Batteries? They're all about redox reactions. That phone you're probably reading this on? Powered by the controlled chaos of electrons zipping back and forth.
What about something a bit more… edible? When you toast a piece of bread, you're actually causing some pretty cool redox reactions. The sugars and other molecules in the bread are getting oxidized, turning that pale dough into a warm, toasty delight. It’s like a delicious, slow-burn fireworks display happening within the bread’s structure. Just don’t burn it, or you get a whole different kind of spectacular, but less tasty, reaction.
Putting It All Together: The Worksheet Wonderland
So, when you’re staring down that dreaded "Acid-Base Precipitation and Redox Reactions Worksheet," don't panic. It’s not some alien language designed to torment you. It's just a way for you to practice identifying these common chemical dramas.
Let’s break down what you might see. You’ll probably have scenarios where you have to:
- Identify the reactants: These are the players entering the chemical arena. Think of them as the opening acts of a concert.
- Predict the products: What’s going to happen when these guys meet? Will they form a solid precipitate? Will they neutralize each other? Will electrons start flying around like confetti at a wedding?
- Determine if it’s an acid-base, precipitation, or redox reaction (or a combination!): Sometimes, these reactions are like a chemistry love triangle, all happening at once.
- Write balanced chemical equations: This is like making sure everyone who showed up to the party gets a name tag and a seat. Every atom needs to be accounted for.
Imagine you’re a detective at a crime scene, but instead of fingerprints, you're looking for evidence of electron transfer or the formation of a solid. Did the suspect leave behind a powdery residue (precipitation)? Did they get electrocuted (redox)? Or perhaps they just had a really intense argument with a strong personality (acid-base)?
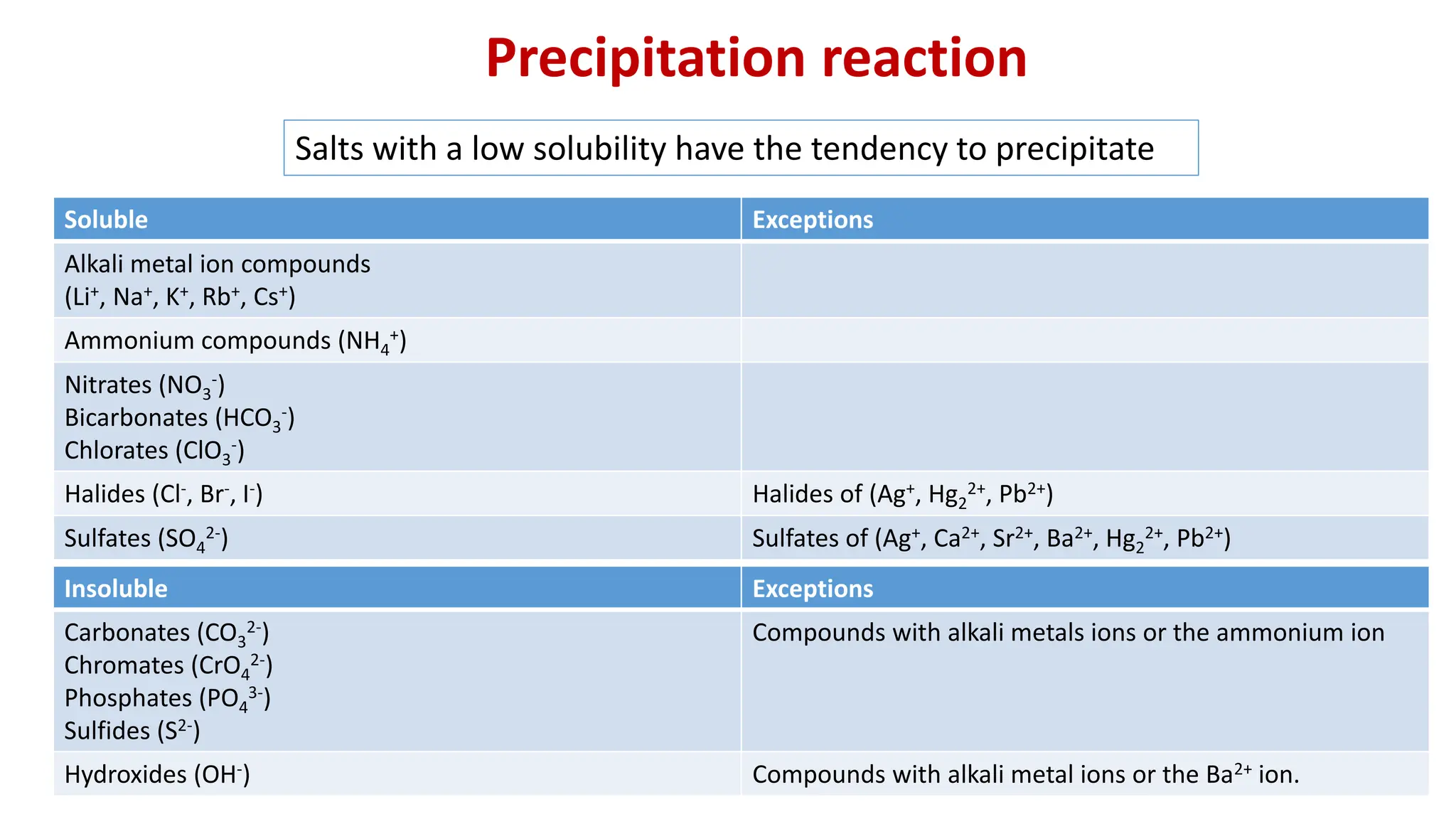
For precipitation, look for clues like "insoluble," "solid forms," or the presence of common ionic compounds that are known to be sparingly soluble. Think of it as spotting a suspiciously large lump in your smooth soup. For acid-base, look for acids (those ending in -ic or -ous, or starting with H+) and bases (those ending in -hydroxide or -amine, or containing OH-). They’re like the sugar and the lemon in your tea – they react to make something different.

And for redox? This is where you gotta be eagle-eyed. Look for changes in oxidation states. If an element’s oxidation state goes up, it’s lost electrons (oxidized). If it goes down, it’s gained electrons (reduced). It’s like checking someone’s bank account: did it go up (gain) or down (loss)? Often, you’ll see elements in their elemental form (like O2, Na, Fe) reacting with compounds, which is a big hint that electrons are about to go on an adventure.
Consider the humble act of making scrambled eggs. You crack the egg, and that liquid goo is a marvel of chemistry. When you heat it up, the proteins in the egg undergo a change. This is a form of oxidation, essentially "cooking" the egg. The molecules are being rearranged and altered by heat, which is like a less direct form of electron transfer. The proteins denature, changing their structure from a floppy, dissolved state to a more rigid, solid state. It’s like your favorite comfy sweater suddenly deciding it wants to be a suit of armor.
Or how about the simple act of taking a vitamin C tablet? Vitamin C is a powerful antioxidant. What does that mean? It means it’s really good at donating electrons to unstable molecules (free radicals) in your body, neutralizing them before they can cause damage. It's like a tiny, molecular superhero swooping in to save the day by giving away its power. That’s a redox reaction happening right inside you, all thanks to a little citrusy compound.

Even the way your body digests food involves a complex symphony of chemical reactions, many of which are redox. Enzymes act as catalysts, speeding up these processes. When you eat food, your body breaks it down, extracting energy. This energy extraction is often achieved through controlled oxidation of molecules like glucose. It’s a slow, efficient burn, very different from lighting a match, but the principle of energy release through electron transfer is the same.
So, next time you see a worksheet with "Acid-Base Precipitation and Redox Reactions" emblazoned across the top, don’t let it intimidate you. Think of it as your chance to become a master of the mundane, a connoisseur of chemical quirks. You’re not just solving problems; you’re understanding the hidden chemistry behind everyday phenomena, from the fizz of your soda to the rust on your bike. You’re basically a kitchen chemist, a bathroom alchemist, and a body mechanic, all rolled into one. And that, my friends, is pretty darn cool.
Remember, every reaction is a story. Precipitation is a breakup, redox is a fiery romance (or feud!), and acid-base is the awkward first date. Just follow the clues, identify the players, and you’ll be solving these chemical conundrums like a pro. And who knows, you might even start seeing the world through a slightly more reactive, and a lot more interesting, lens. Now go forth and conquer that worksheet! Your inner chemist awaits!
