Acetic Acid And Sodium Hydroxide Balanced Equation

Hey there, science curious folks! Ever wondered what happens when you mix a couple of common household ingredients and things get all… balanced? Today, we're diving into something that sounds a bit fancy but is actually super relatable: the balancing act between acetic acid and sodium hydroxide.
Now, before you picture a mad scientist in a lab coat, let's just chill. Think of it like making a really good cup of tea, or maybe even baking a cake. There's a little bit of science involved, sure, but it's all about getting the right ingredients in the right amounts to create something new and interesting. So, what are these mysterious players in our little chemical drama?
Meet Our Reactants: The Dynamic Duo
First up, we have acetic acid. You probably know it by another name: the main ingredient that gives vinegar its tangy kick! Yep, that stuff you put on your fries or use to clean your windows? That's acetic acid. It's an acid, which means it's got a bit of a sour personality, always looking to share its hydrogen atoms.
Then, we have sodium hydroxide. This one is a bit more of a powerhouse. You might know it by names like lye or caustic soda. It's a strong base, meaning it's the opposite of an acid. Think of it as the generous one, always ready to accept those hydrogen atoms. It's used in things like soap making and drain cleaners, so it's definitely got some muscle.
The Big Question: What Happens When They Meet?
So, what happens when this tangy acid and this strong base decide to hang out? Well, they have a little chemical party, and the main event is a neutralization reaction. It's like when two people with totally opposite personalities meet and somehow end up being best friends because they balance each other out perfectly.
When acetic acid and sodium hydroxide meet, they don't just say hello and move on. They actually react with each other. The acid gives up a hydrogen ion (H+), and the base happily scoops it up. This might sound super technical, but think of it like this: the acid has something it wants to get rid of, and the base is perfectly designed to take it.
This little transfer changes both of them. The acetic acid, after giving away its hydrogen, becomes acetate. And the sodium hydroxide, after accepting that hydrogen, becomes water. Pretty neat, right? They're essentially transforming into something new and, in this case, much more stable and less reactive.
The Magic of the Balanced Equation
Now, here's where the "balanced equation" part comes in. In chemistry, we like to be super precise. We don't just want to know what happens, but how much of each thing is involved. It’s like a recipe – you don’t just throw in random amounts of flour and sugar, right? You want the right proportions for the best result.
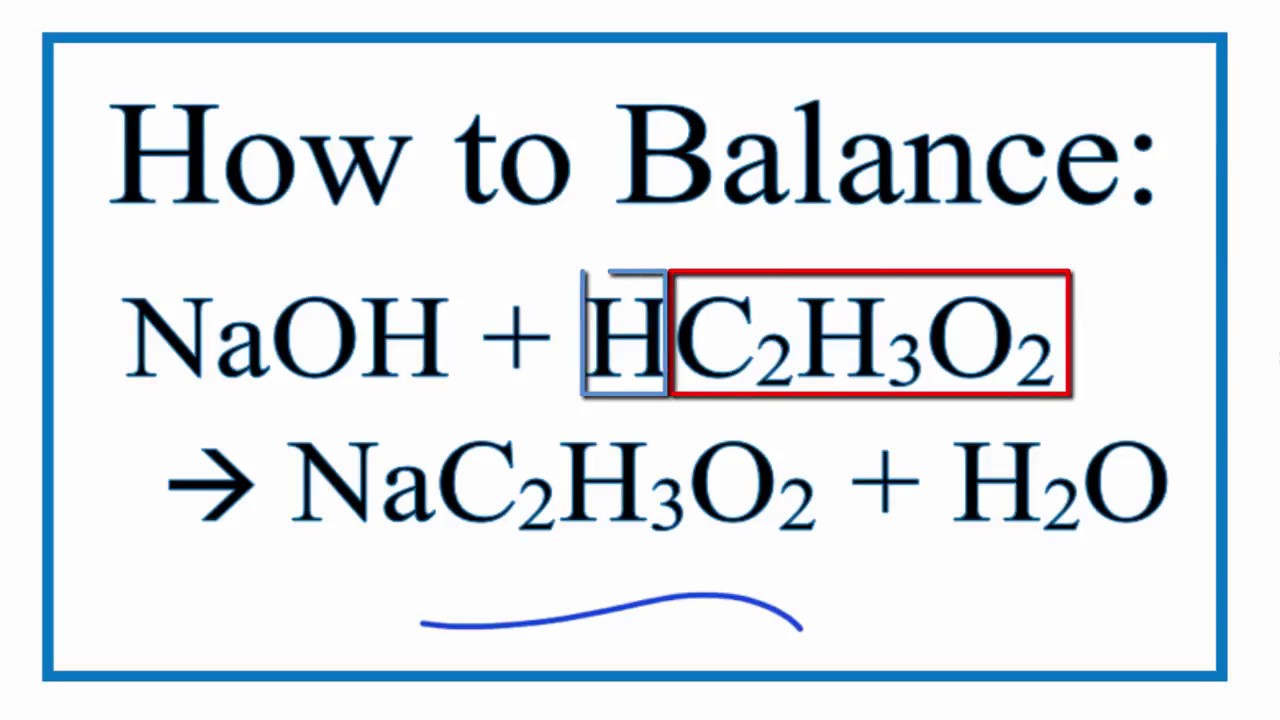
The balanced equation for this reaction looks like this:

CH₃COOH (acetic acid) + NaOH (sodium hydroxide) → CH₃COONa (sodium acetate) + H₂O (water)
Let's break that down. CH₃COOH is our vinegar friend, acetic acid. NaOH is our strong base, sodium hydroxide. On the other side of the arrow, we have CH₃COONa, which is sodium acetate. This is the salt formed from the acid and base. And H₂O? That's good old water!
Why is "Balanced" So Important?
The word "balanced" here is the key. It means that the number of atoms of each element on one side of the equation (the "reactants" side, where our original ingredients are) is exactly the same as the number of atoms of that element on the other side (the "products" side, where our new stuff is).
Look closely at the equation. We have:

- Carbon (C) atoms: 2 on the left, 2 on the right.
- Hydrogen (H) atoms: 4 on the left (3 in CH₃ and 1 in COOH) + 1 in NaOH = 5. On the right: 3 in CH₃ + 2 in H₂O = 5.
- Oxygen (O) atoms: 2 in CH₃COOH + 1 in NaOH = 3. On the right: 2 in CH₃COONa + 1 in H₂O = 3.
- Sodium (Na) atoms: 1 on the left, 1 on the right.
See? Everything matches up! This is a fundamental principle in chemistry called the Law of Conservation of Mass. Basically, matter can't be created or destroyed, it just changes form. So, all the atoms that went into the reaction have to come out, just rearranged into new molecules.
Think of it like LEGOs. You start with a pile of red and blue bricks, and you build a car. The number of red bricks and blue bricks you started with is the same number you have in the car, they're just put together differently. The balanced equation is just our way of showing that LEGO build in chemical language.
Beyond the Equation: What's the Big Deal?
So, we’ve got acetic acid turning into sodium acetate and water with the help of sodium hydroxide. Cool, but why should we care? Well, this kind of reaction is happening all around us, and it's pretty darn useful!
For starters, this reaction is how we make many common salts. Sodium acetate, for example, has a few cool uses. It can be used as a food additive (E262), acting as a preservative and a flavor enhancer. It's also used in things like hand warmers, where a controlled crystallization process releases heat!
And the fact that it neutralizes acids is super important. In our bodies, for instance, our stomach acid (which is hydrochloric acid, a different kind of acid) needs to be balanced. Sometimes, we take antacids, which are bases, to neutralize excess stomach acid. It's a similar principle, just with different players.
A Friendly Chemical Exchange
The reaction between acetic acid and sodium hydroxide is a perfect example of how opposites attract in chemistry and create something new and often, quite useful. It’s a simple exchange, a friendly chemical handshake where the acid and base find common ground, resulting in a salt and water.
It’s a reminder that even everyday substances have fascinating chemical stories to tell. And understanding these simple reactions, like our balanced equation here, is like getting a peek behind the curtain of how the world works. So next time you’re enjoying some vinegar, or perhaps even making soap, give a little nod to the amazing, balanced world of chemistry!
