According To Table F Which Ions Combine With Chloride
Ever find yourself staring at a list of elements, feeling like you’ve stumbled into a secret language lesson from a grumpy wizard? Yeah, me too. Specifically, that moment when someone casually mentions "Table F" and you’re suddenly transported back to chemistry class, conjuring images of dusty textbooks and the faint scent of ethanol. But fear not, my friends! Today, we’re not diving into the murky depths of ionic bonding with a textbook definition that makes your eyes glaze over. Instead, we’re going to chat about chloride ions, those little guys who are just itching to team up with other elements, and we’re going to do it in a way that’s as breezy as a summer picnic.
Think of chloride ions like the social butterflies of the elemental world. They’re constantly looking for a dance partner, a buddy to hang out with. And who can blame them? Being a lone ion, floating around by yourself, can get a bit… well, lonely. It’s like being the last person at a party, desperately trying to make conversation with the leftover chips. Not exactly thrilling, is it?
So, who are these chloride ions looking to mingle with? This is where our trusty (and sometimes baffling) "Table F" comes into play. Now, I’m not going to make you memorize the whole thing. That’s like asking me to remember my PIN number while I’m simultaneously trying to parallel park on a busy street – a recipe for disaster. Instead, let’s focus on the vibe, the general crowd that chloride ions tend to flock towards.
Imagine Table F as a giant matchmaking service for elements. It’s got profiles, compatibility scores, and maybe even a few cheesy pickup lines. Chloride ions, denoted as Cl⁻ (don’t worry, we’ll unpack that little minus sign in a bit), are essentially looking for someone who’s feeling a little bit… positive. Like a perfectly ripe avocado, they’re just waiting for that right touch to complete the experience. They’re the yin to someone else’s yang, the peanut butter to their jelly, the… you get the idea.
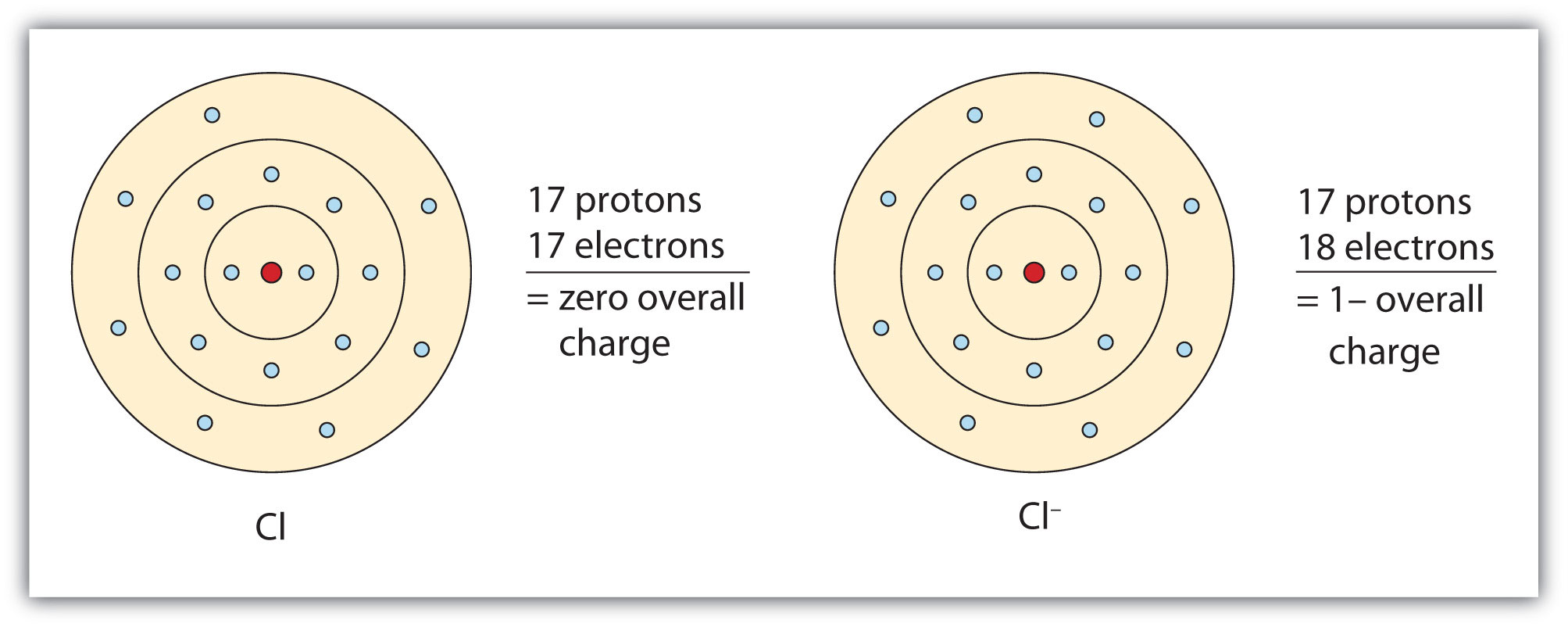
The minus sign next to that ‘Cl’? That’s the key! It means our chloride friend has an extra electron, a little something they’re eager to share or, more accurately, attract. Think of it like having an extra coupon for a free ice cream cone. You’re not going to keep that to yourself, are you? You’re going to find someone who’s craving a scoop of happiness, and in the element world, that someone usually has a positive charge. They’re like the person who loves sharing their Netflix password – everyone’s a winner!
So, who are these positive folks? According to the wisdom of Table F (which, let’s be honest, is a bit like a cryptic crossword puzzle written by a particularly enthusiastic chemist), chloride ions have a major crush on certain types of elements. We’re talking about the ones that are positively charged, or in chemistry terms, are cations. These are the elements that have lost electrons, making them feel a bit bare and longing for a friend. They’re the ones who accidentally sent that text to the wrong person and are now desperately trying to recover. Chloride, with its extra electron, is like the calm, collected friend who swoops in to save the day.
The Usual Suspects: Alkali and Alkaline Earth Metals
The most common companions for our chloride pals are usually found lurking in the first couple of columns of the periodic table. These are your alkali metals (Group 1) and alkaline earth metals (Group 2). Think of them as the popular kids at the elemental prom, always looking for a good time and a stable partner. They’re predictable, reliable, and generally a pretty safe bet.

Take Sodium, for example. That’s Na, if you’re feeling fancy. Sodium is a Group 1 element, and it’s always ready to ditch one of its electrons. It’s like it has too many socks and is just looking for someone to give one to. When sodium meets chloride, BAM! You get Sodium Chloride. Yep, that’s your everyday table salt. See? We told you this was relatable!
Imagine you’re making popcorn. You’ve got the kernels, you’ve got the butter, and then you reach for the salt. That little sprinkle of NaCl? That’s a happy couple, a perfect union of sodium and chloride ions, all thanks to the magic of electrostatics and, you know, Table F giving them the green light.
Then there’s Potassium (K), another Group 1 superstar. Potassium is a bit like sodium’s slightly more athletic cousin. It also likes to lose an electron. Pair it up with chloride, and you get Potassium Chloride (KCl). This stuff is sometimes used as a salt substitute, because even when they’re not making the popcorn we all love, these guys are still teaming up.
Moving over to the alkaline earth metals, you’ve got elements like Magnesium (Mg) and Calcium (Ca). These guys are in Group 2, which means they’re a little bit more cautious. They’re like the friends who bring a plus-one to the party, but they’re willing to share. Magnesium, for instance, likes to lose two electrons. So, it’s like it has two extra socks it needs to offload. When it meets chloride, it’s going to grab two chloride ions to keep things balanced. Think of it as a double date! Magnesium Chloride (MgCl₂) is what you might find in some de-icing salts or even as a supplement. It’s all about finding the right balance, like a perfectly proportioned smoothie.

Calcium is another big player here. When calcium, with its two positive charges (it’s lost two electrons!), meets up with chloride ions, you get Calcium Chloride (CaCl₂). This is the stuff that’s a champion at melting ice and keeping dust down on roads. It’s a bit of a workhorse, and it’s all thanks to calcium’s eagerness to lose electrons and chloride’s willingness to grab them. It’s like a construction crew and a demolition team working in perfect harmony, albeit on a microscopic scale.
Don’t Forget the Transition Metals!
Now, the periodic table isn’t just rows and columns; it’s also got those funky middle sections, the transition metals. These guys are the rebels of the chemical world. They’re a bit more… unpredictable. Sometimes they’ll lose one electron, sometimes they’ll lose two, sometimes even more! They’re like that friend who changes their mind every five minutes about what movie to watch. This variability can make them a little trickier to pin down in a neat little box, but chloride ions are still very happy to dance with them.
Think about Iron (Fe). Iron is famously known for rusting, which is basically iron reacting with oxygen and water. But iron also forms compounds with chloride. Depending on the situation, iron can exist as Fe²⁺ (meaning it’s lost two electrons) or Fe³⁺ (it’s lost three electrons). So, you can have Iron(II) Chloride (FeCl₂) or Iron(III) Chloride (FeCl₃). It’s like Iron showing up to the chloride party with either two or three of its coolest accessories. Chloride is just happy to hang out, no matter the vibe.
Copper (Cu) is another example. It can be Cu⁺ or Cu²⁺. So, you can get Copper(I) Chloride (CuCl) or Copper(II) Chloride (CuCl₂). These compounds are used in various applications, from pigments to agriculture. It’s proof that even these somewhat temperamental metals find a stable partner in chloride. They’re like the friend who’s always a little dramatic, but you love them anyway, and chloride is the steady influence.
The key here is that these transition metals have a positive charge, making them attractive to the negatively charged chloride ion. Table F, in its infinite wisdom, lays out these possibilities, showing us that chloride isn’t just picky; it’s actually quite versatile. It’s the ultimate connector, the element equivalent of a party planner who knows how to get everyone mingling.
Why Do They Combine? The Magnetic Pull of Opposite Charges
So, why this constant pairing? It all comes down to a fundamental principle in chemistry: opposites attract. Remember that little minus sign on the chloride ion? That means it has a negative charge. The metals we’ve been talking about, the alkali metals, alkaline earth metals, and transition metals, all tend to form positive ions. They have a positive charge.
It’s like putting two magnets together. You know how if you hold the north pole of one magnet near the south pole of another, they snap together with surprising force? That’s pretty much what’s happening here, on an atomic level. The positive charges of the metal ions are strongly attracted to the negative charge of the chloride ion. This attraction is what holds them together, forming a stable compound. It’s the universe’s way of saying, “Hey, you two seem to get along, why don’t you stick together?”
This electrostatic attraction is super important. It’s what forms ionic bonds. These are the strong glue that holds these compounds together. Without these bonds, sodium and chlorine would just be individual elements, potentially causing all sorts of chaos. But together, as NaCl, they’re stable, useful, and, in the case of salt, downright delicious.

Table F: The Cheat Sheet to Chemical Compatibility
Now, let’s circle back to Table F. What is it, really? It’s not a dating app, but it does give us clues about which elements are likely to form stable partnerships. While Table F primarily focuses on solubility (whether something dissolves in water), it indirectly tells us about the types of ions that tend to form stable compounds. Generally, if a chloride compound is listed as insoluble in water, it means the ions involved are holding on very tightly to each other, forming a strong, stable lattice. If it's soluble, it means water can come between the ions relatively easily.
For our purposes today, the key takeaway is that chloride ions are highly likely to combine with any element that readily forms a positive ion. These are overwhelmingly metals. The specific types of metals and the exact way they combine are detailed in more advanced chemistry, but for a general understanding, focusing on the positively charged metals is your best bet.
Think of Table F as a celebrity gossip magazine for elements. It tells you who’s hanging out with whom, who’s a hot commodity, and who’s just there for the free snacks. Chloride is always at the party, and it’s usually seen with the popular metallic crowd.
So, the next time you hear about Table F and chloride ions, don’t panic. Just picture those social butterflies, the chloride ions, eagerly seeking out their positive counterparts. From the salt on your dinner table to the de-icing agents on the roads, these ionic friendships are all around us, making the world a more stable, and often tastier, place. They’re the unsung heroes of chemical bonding, proving that sometimes, all it takes is a little bit of attraction to create something truly remarkable.
