According To Table F Which Compound Is Soluble In Water

Hey there, fellow curious minds! Ever found yourself staring at a bunch of chemical names and wondering, "Which one of these guys is gonna play nice with water?" It's a surprisingly common question, whether you're a budding scientist in a lab coat, a home cook trying to dissolve something for a recipe, or just someone who spilled a mystery liquid and is hoping for the best. Today, we're going on a little adventure to explore just that, with a little help from a handy tool called Table F.
Now, "Table F" might sound a bit… formal, right? Like something you’d find in a dusty textbook. But trust me, it's actually one of the coolest little cheat sheets in chemistry. Think of it like a restaurant menu for solubility. You know, where you can glance at the dishes and see which ones are "gluten-free" or "vegetarian"? Table F is kind of like that, but instead of dietary needs, it tells you if a compound is going to dissolve in water, or if it's going to be that stubborn ingredient that just floats on top. Pretty neat, huh?
So, why is this whole "dissolving in water" thing such a big deal? Well, water is, you know, pretty much everywhere. It's in us, it's in the rain, it's in our tea. And for lots of chemical reactions to happen, things need to be able to move around and bump into each other. When a substance dissolves in water, it breaks apart into tiny little pieces – we call them ions – that can then happily mingle with the water molecules. It's like a big, splashy party where everyone's invited and can dance freely!
Imagine trying to make a refreshing lemonade. You've got your water, and you want to add some sugar, right? If the sugar didn't dissolve, you'd just have a gritty mess at the bottom of your glass. Not exactly a thirst quencher. Or think about laundry detergent. It needs to dissolve in the wash water to grab onto all those pesky dirt particles. Without solubility, our lives would be a lot more… lumpy.
Okay, so back to Table F. What does it actually tell us? It's usually a list of different types of compounds and a general rule about their solubility in water. It’s not just a random assortment of chemicals; it’s organized by families or categories. Think of it like grouping your toys: you have your LEGOs here, your stuffed animals there. Table F groups chemicals based on what they're made of.
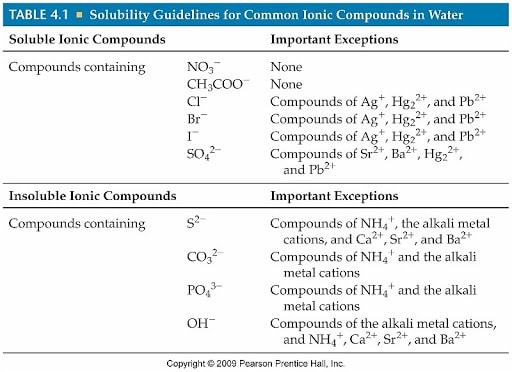
The most common categories you'll see on Table F relate to salts. Now, "salt" in chemistry isn't just the stuff you sprinkle on your fries. It's a broader term for compounds formed when an acid and a base react. They're often made of a metal atom and a non-metal atom. And the rules on Table F help us predict if these salts are going to be water-lovers or water-haters.
The Big Guns: Usually Soluble!
There are some groups of compounds that Table F pretty much guarantees will dissolve in water. These are your VIPs of solubility, the life of the aquatic party! For instance, any compound containing an alkali metal is a safe bet. Alkali metals are things like lithium (Li), sodium (Na), potassium (K), and so on. Ever heard of sodium chloride, aka table salt? That's sodium (an alkali metal!) and chlorine. And guess what? It dissolves in water like nobody's business. You could build a whole salt fort in the ocean, and it would eventually just… melt away.
Another super-soluble crew are compounds containing the ammonium ion (NH₄⁺). If you see that little gang hanging out with something else, chances are they're heading for a watery embrace. Ammonium compounds are used in all sorts of things, from fertilizers to cleaning products, and their ability to dissolve is key to how they work.

What about other common ions? Compounds containing nitrates (NO₃⁻) and acetates (CH₃COO⁻) are also almost always soluble. So, if you see something like potassium nitrate or sodium acetate, you can be pretty confident they'll be happily dissolved in your water. It’s like having a golden ticket to the solubility express!
The Tricky Bunch: It Depends!
Now, things get a little more interesting when we move to other groups. For example, compounds containing chlorides (Cl⁻), bromides (Br⁻), and iodides (I⁻) are generally soluble. But here’s the catch: there are a few exceptions. Silver (Ag⁺), lead (Pb²⁺), and mercury (Hg₂²⁺) ions hate water when they're hanging out with these halides. So, while most chlorides dissolve, silver chloride? Nope, it forms a solid that sinks to the bottom. It's like a bouncer at the water party, saying "You're not on the list!"

Sulfates (SO₄²⁻) are another group that are mostly soluble. But again, there are some notorious exceptions. Barium (Ba²⁺), strontium (Sr²⁺), calcium (Ca²⁺), lead (Pb²⁺), silver (Ag⁺), and mercury (Hg₂²⁺) ions will form insoluble sulfates. So, if you see a sulfate with one of these metals, expect it to stay solid. It's a bit like how in a big group of friends, there are always a few who prefer to sit out the dance.
The "Nope" List: Usually Insoluble
Then you have the compounds that are generally insoluble in water. These are your hydrophobic characters, the ones who prefer to stay away from the H₂O. This category often includes compounds containing carbonates (CO₃²⁻), phosphates (PO₄³⁻), sulfides (S²⁻), and hydroxides (OH⁻).
However, and you’re probably seeing a pattern here, there are always exceptions! Carbonates, phosphates, and sulfides are usually insoluble unless they are paired with an alkali metal cation (like sodium or potassium) or the ammonium ion. So, calcium carbonate (think of chalk or seashells) is insoluble. But sodium carbonate (washing soda)? That dissolves just fine! It’s like some people are naturally aloof, but they’ll warm up if they're with their best buddies.
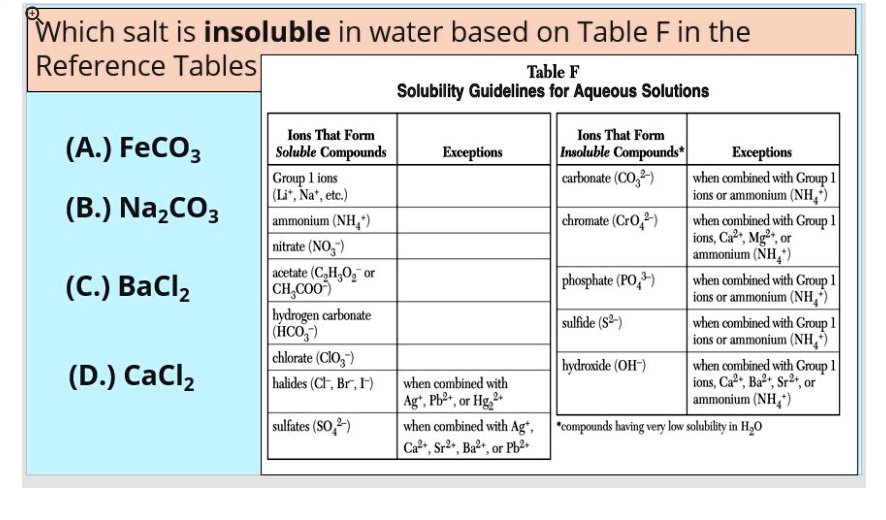
Hydroxides (OH⁻) are a bit of a mixed bag. Most are insoluble, but the hydroxides of alkali metals (sodium hydroxide, potassium hydroxide – these are strong bases!) and the heavier alkali earth metals like calcium, strontium, and barium are actually soluble. So, while iron(III) hydroxide (rust-ish!) is insoluble, barium hydroxide is soluble. It’s a bit of a chemistry curveball!
So, when you’re looking at Table F and asking yourself, "Which compound is soluble in water?", you’re essentially looking for the compound that fits into one of the "generally soluble" categories and doesn't fall into one of the listed exceptions. It’s like solving a little logic puzzle!
It’s fascinating how these simple rules can predict the behavior of so many different substances. It’s the backbone of understanding everything from how medicines dissolve in our bodies to how minerals form in the Earth. So next time you’re wondering about solubility, remember Table F. It’s not just a table; it’s a little window into the watery world of chemistry, and it’s pretty cool to be able to peek through!
