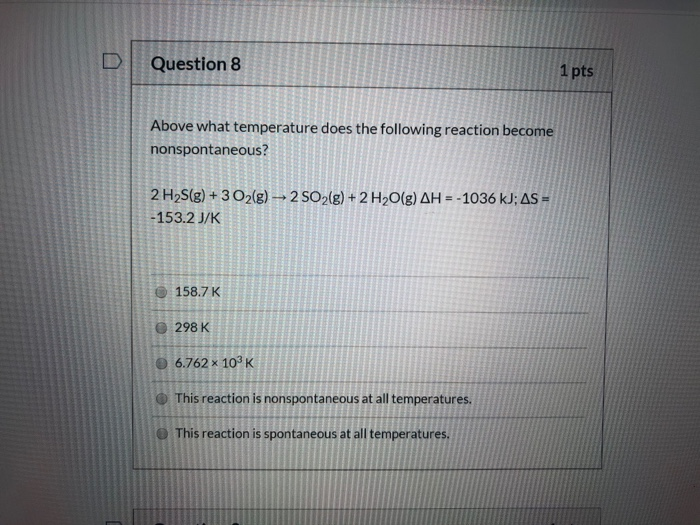
Above What Temperature Does The Following Reaction Become Nonspontaneous
Ever wondered why some things just happen on their own, while others need a good push? It's like magic, but it's actually science! And today, we're diving into a fun bit of chemistry that explains it all: spontaneity. This isn't just for scientists in lab coats; understanding when a reaction will (or won't!) happen can be surprisingly useful and even a bit of a party trick!
So, what exactly is a spontaneous reaction? Think about a ball rolling downhill – it just goes! That's spontaneous. A reaction that requires energy to happen, like pushing that ball uphill, is nonspontaneous. The "magic number" we're after is a temperature threshold. Above a certain point, a reaction that was once unwilling to go becomes eager to start, all on its own. This is a fundamental concept in chemistry, and it touches everything from baking a cake to how our bodies work.
For beginners, this concept is a fantastic introduction to the forces that govern chemical change. It helps demystify why certain processes occur and others don't. Families might find it fascinating to relate this to everyday cooking! For instance, why does baking soda and vinegar fizz up immediately (spontaneous!) but a cake needs heat to bake (requiring energy input, but becomes spontaneous at baking temperatures)? Hobbyists, like gardeners, can even think about soil decomposition – some parts break down easily, while others are more stubborn, influenced by temperature and other factors.
Let's look at a specific example, though we won't get into scary math. Imagine a reaction where the entropy (think of it as the messiness or disorder) increases significantly. At low temperatures, the energy released or absorbed (enthalpy) might be the bigger player, dictating spontaneity. But as you crank up the heat, that increase in entropy starts to win out. So, for a reaction with a large positive entropy change, there's a temperature above which it will become spontaneous. It’s like a party – at first, everyone's a bit reserved, but as the music gets louder (higher temperature!), more people join in the dancing (spontaneous reaction!).
Now, for the million-dollar question: above what temperature does a specific reaction become nonspontaneous? This is where a little bit of information comes in handy! We need to know two key things about the reaction: its enthalpy change (ΔH – how much heat it absorbs or releases) and its entropy change (ΔS – how much its disorder changes). The magic equation, for those who are curious, is roughly: Temperature (T) = -ΔH / ΔS. If ΔH is positive (the reaction absorbs heat, like melting ice) and ΔS is positive (it gets messier, like ice melting into water), there will be a temperature where it flips from nonspontaneous to spontaneous. If ΔH is negative (releases heat) and ΔS is negative (gets more ordered), it will also have a temperature flip, but in the other direction.

Getting started with this idea is easy! Next time you're cooking, think about the ingredients and the heat. Why does sugar caramelize when heated? Why does an egg cook? Observe these changes and ponder what forces are at play. You don't need a lab to appreciate the elegance of chemical spontaneity!
Understanding when reactions are spontaneous or not is a little window into the fundamental workings of the universe. It’s a concept that bridges the gap between theoretical science and our everyday experiences, making the world around us a little more understandable and a lot more interesting. So go forth, and observe the spontaneous wonders around you!
