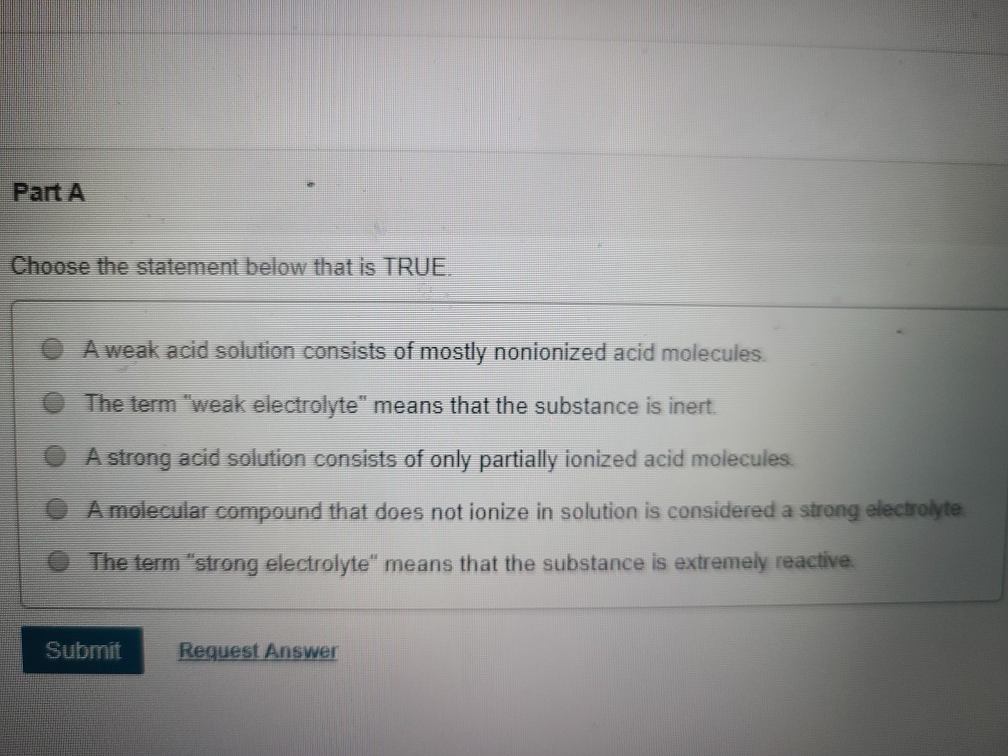
A Weak Acid Solution Consists Of Mostly Nonionized
Imagine your favorite sweet treat, perhaps a perfectly ripe strawberry or a zesty lemon tart. They owe their delicious tang to something called a weak acid. You might think of acids as something scary, like something that could melt through a sink! But the acids in our food are usually the gentle, mild kind.
And here's a little secret about these friendly food acids: they're a bit shy. When you mix them into water, like when you make lemonade or squeeze those strawberries, most of them decide to stay just as they are. They're like little introverts at a party, preferring to hang out in their own little groups rather than mingle with everyone else.
This preference for staying "nonionized" is what makes them weak. Think of "ionized" as breaking apart and becoming a bit more… energetic. Our weak acids are content just being themselves, mostly unbothered and still perfectly capable of adding that delightful sour note.
Let's take acetic acid, the main ingredient that gives vinegar its punch. When you put vinegar in your salad dressing, most of the acetic acid molecules are perfectly happy sticking together. They’re like a group of friends who just want to chat amongst themselves.
Only a tiny fraction of them decide to break off and become "ions." These ions are a bit more active, and they're the ones who do most of the sour-tasting work. But it's a small crowd, really! The vast majority are still enjoying their un-ionized existence.
It's a bit like a family reunion. You have the core family members, who are very close and stay together. Then you might have a few cousins who are visiting from out of town and are a bit more adventurous. But the heart of the gathering is the main family unit, right?
This is why we call them "weak." They don't readily split up into their charged components. They're not as eager to dive into the molecular fray as their stronger, more boisterous acid cousins.
![[ANSWERED] Choose the statement below that is TRUE. The term "weak - Kunduz](https://media.kunduz.com/media/sug-question/raw/53161055-1659276472.2016394.jpeg?h=512)
Think about citric acid in oranges or limes. It's what makes them so refreshingly tart. When you squeeze a lime, the citric acid is mostly in its whole, un-ionized form, just chilling in the juice.
A few molecules might break apart, but it's a modest number. They're not causing a huge commotion. They're just adding their little bit of zing, and the bulk of the citric acid is still very much intact.
It’s rather like a quiet hum versus a loud shout. The weak acid solution is a gentle hum of sourness, largely thanks to the majority of its molecules choosing not to ionize.
This has some surprisingly practical and heartwarming implications. Because weak acids are mostly un-ionized, they are generally much gentler. They won't corrode things as readily as strong acids do.

This is why they're safe to have in our food and drinks. Imagine if the citric acid in your orange juice decided to go all out and ionize completely! Your stomach might not be too happy about that. Thankfully, its weak nature keeps it from being too aggressive.
It's a beautiful balance, really. The small amount of ionization provides that desirable sour taste, while the overwhelming presence of nonionized molecules keeps things safe and pleasant.
Consider the humble yogurt. The sourness you taste comes from lactic acid, another weak acid. It's produced by bacteria, and it's mostly in its nonionized form.
This makes yogurt a fantastic food. It's tangy and delicious, but also gentle on your digestive system. The lactic acid is there to give it flavor, but it's not aggressively attacking anything.

The "mostly nonionized" aspect is like a built-in safety feature. It’s the acid’s way of saying, "I’m here to add some flavor, but I promise not to cause too much trouble."
It’s almost as if the weak acid is trying to be considerate. It knows its job is to add zest, but it doesn't want to overwhelm the delicate balance of your taste buds or your stomach.
So, the next time you enjoy a sour candy, a tangy salad dressing, or a refreshing glass of lemonade, take a moment to appreciate the unsung heroes: the mostly nonionized molecules of weak acids!
They're like the quiet, dependable friends who are always there to add a little something special, without being overbearing. They're the foundation of many delicious flavors, and their gentle nature is a gift.

It's a fascinating microscopic world. Billions upon billions of molecules, and most of them are just happily existing in their original form, contributing to the simple pleasures of life.
The strength of an acid isn't about how loud or aggressive it is, but rather how willing it is to change its form. Weak acids are content with their current state, and that's what makes them so special and so useful to us.
It’s a testament to nature's clever design. We get the flavor we love, and the acids do their job in a way that’s perfectly suited for our enjoyment and well-being.
So, a round of applause for the shy, the gentle, and the wonderfully nonionized weak acids! They’re the unsung flavor-makers in our everyday lives, and they deserve a little recognition for their subtle yet significant contributions.
They prove that being "weak" doesn't mean being ineffective. It just means being present in a way that’s perfectly balanced and wonderfully mild. Cheers to the un-ionized majority!
