A True Statement About Hydrophobic Interactions Is That They

Hey there, curious minds! Ever wonder what makes oil and water just… not friends? Like, really, really not friends? It's one of those things we all just accept, right? But behind that simple observation lies a super cool scientific concept called a hydrophobic interaction. And guess what? It’s basically a fundamental part of how life itself works. Pretty neat, huh?
So, what's the deal with "hydrophobic"? "Hydro" obviously means water, and "phobic" means, well, fear. So, hydrophobic literally means "water-fearing." Makes sense, right? Things that are hydrophobic don't like to hang out with water. They'd rather be doing their own thing, far away from the H₂O crowd. Think of it like that one friend at a party who just hates the dance floor. They'd much rather chill in the quiet corner with their other non-dancing pals.
But here's the kicker: this "fear" isn't an active emotion like you or I might feel. It's more of a preference, a consequence of how these molecules are built. We're talking about molecules here, those tiny building blocks of everything around us. Some molecules are like little magnets for water, they’re called hydrophilic. Others? They’re the opposite. They’re the wallflowers of the molecular world when it comes to water.
Why is this a true statement? Because it's not just a theory; it's a fundamental observation of how certain substances behave. It’s a consistent pattern we see again and again. Imagine trying to mix oil into water. You stir and stir, and what happens? It just separates, right? The oil molecules huddle together, and the water molecules do the same. They’re basically saying, "Nope, not mixing!"
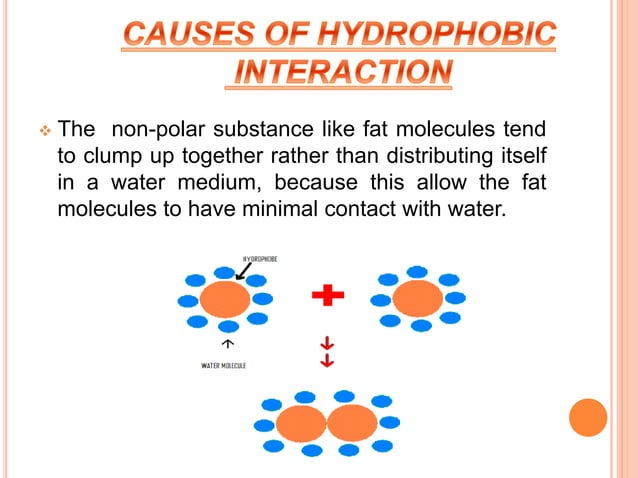
So, why do they do this? It all boils down to something called energetic stability. Basically, the universe likes things to be as "easy" or as stable as possible. Water molecules are super attracted to each other. They form these little clusters, like a group of friends holding hands. When a hydrophobic molecule (like an oil molecule, which is made of long chains of carbon and hydrogen atoms) enters this water party, it disrupts the water's happy little hand-holding. It's like a lone wolf trying to join a tight-knit dance group. It just doesn't fit in comfortably.
To minimize this disruption, the water molecules sort of rearrange themselves. They try to surround the hydrophobic molecule in a way that's less… awkward. But the most stable arrangement for both the water and the hydrophobic molecules is for them to separate. The hydrophobic molecules then group together, minimizing their contact with water, and the water molecules get back to their own happy huddling. This clustering of hydrophobic molecules is the essence of a hydrophobic interaction.
Think of it like this: imagine you're at the beach, and you have a bunch of little bouncy balls (hydrophobic molecules) and a bucket of water. If you throw the bouncy balls into the water, they don't dissolve. They bob on the surface or sink to the bottom, but they don't become one with the water. And what happens to the bouncy balls themselves? They tend to clump together, right? They avoid getting too much water on them by sticking to each other. That's a visual for hydrophobic interaction!
Now, why is this so cool? Because this seemingly simple repulsion is responsible for some of the most fundamental structures and processes in biology. Seriously, life as we know it wouldn't exist without hydrophobic interactions!
Let's talk about cells, the basic units of all living things. Cell membranes, the outer walls of these tiny living factories, are made of molecules called phospholipids. These phospholipids are fascinating because they're like tiny tadpoles. They have a "head" that loves water (hydrophilic) and a "tail" that hates water (hydrophobic).

In the watery environment of our bodies, these phospholipids spontaneously arrange themselves into a double layer, called a bilayer. The hydrophilic heads face outwards, towards the water inside and outside the cell, and the hydrophobic tails all face inwards, away from the water. This creates a protective barrier, a bit like a tiny, molecular moat and castle wall, keeping the cell's insides safe and separate from the outside world. Without hydrophobic interactions, our cells wouldn't have these essential membranes, and we wouldn't be here!
It's like building with LEGOs. You have different types of bricks. Some fit together easily, some need a bit of a push. But when you're building a house, you want specific walls and a roof. The hydrophobic tails are like the bricks you don't want exposed to the rain, so you tuck them away inside, creating a solid structure. The water-loving heads are the bricks that can handle being out in the elements.

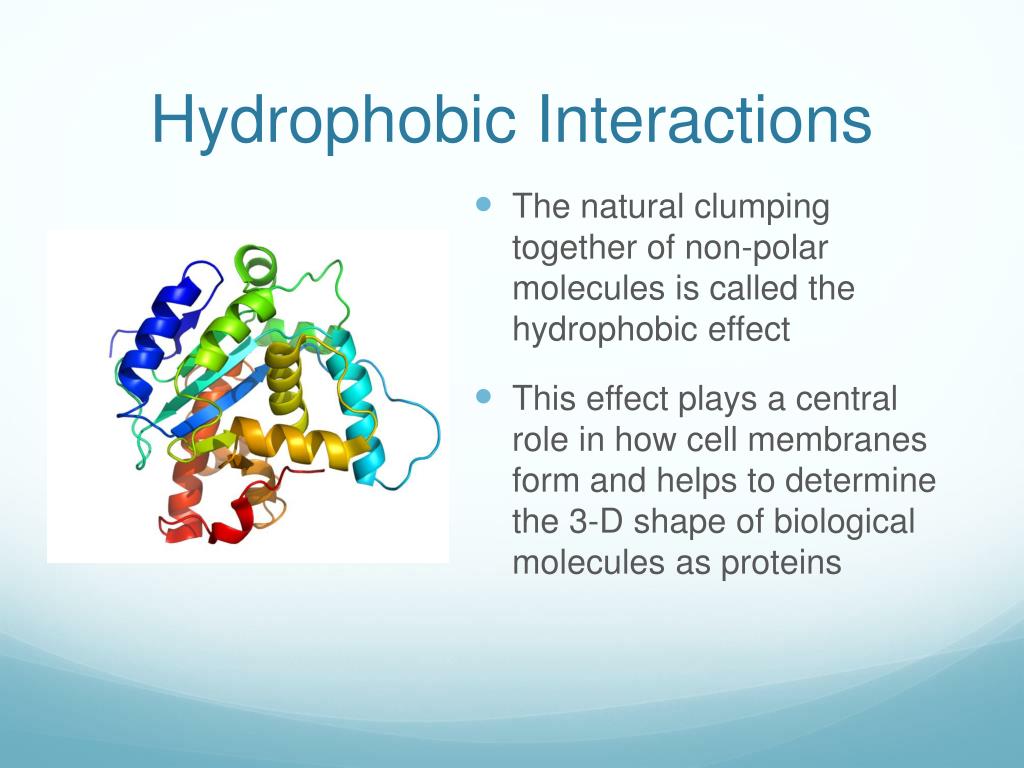
But it doesn't stop there! These interactions are also crucial for how proteins fold into their unique, three-dimensional shapes. Proteins are the workhorses of our cells, carrying out countless jobs. For a protein to function correctly, it needs to be folded in a very specific way. As a protein is being built, the hydrophobic parts of it (those parts that don't like water) tend to get tucked into the interior of the protein, away from the watery surroundings. This "folding away" driven by hydrophobic interactions is a key step in creating a functional protein. It’s like a crumpled piece of paper unfolding into a complex origami shape, with the "ugly" bits hidden on the inside.
Think about a tangled ball of yarn. If you were to try and organize it, you'd naturally pull the looser, more exposed strands inward and create a more compact form. That’s a bit like what happens with proteins, but driven by that water-fearing tendency.
So, next time you're pouring oil and vinegar for a salad dressing and notice them not mixing, or you see a raindrop bead up on a waxy leaf, remember the magic of hydrophobic interactions. It's a true statement about the chemical world, a fundamental principle that’s both incredibly simple in its cause and profoundly complex in its consequences. It’s a silent, invisible force that holds life together. How utterly fascinating is that?
