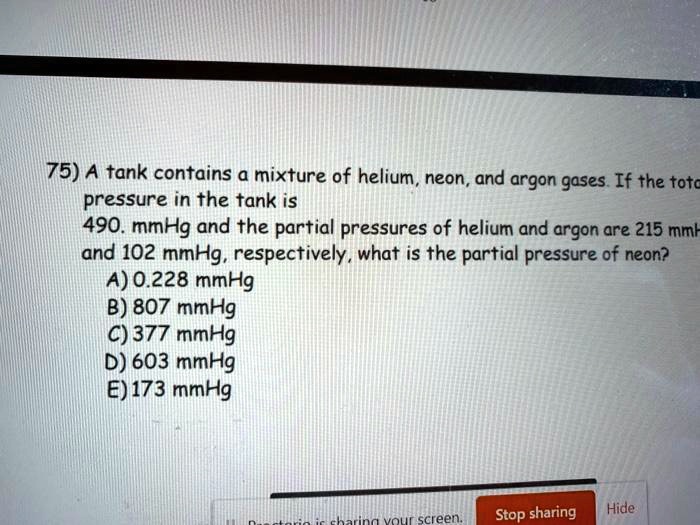
A Tank Contains A Mixture Of Helium Neon And Argon
So, picture this: I’m chilling at a friend’s place, right? He’s a bit of a science nerd, and his apartment is basically a mini-lab. We’re watching some old sci-fi movie, the kind where the aliens are clearly just guys in rubber suits, and suddenly he points to this rather impressive, albeit dusty, tank in the corner. "That," he announces with a dramatic flourish, "is a cocktail of awesome gases."
My initial thought? "Great, he's brewing something illegal again." But no, he explains, it's a mixture of Helium, Neon, and Argon. My eyebrows did that little involuntary arch thing. I’d heard of these gases, of course. Helium in balloons, Neon in those flashy signs, and Argon… well, Argon was the mysterious third wheel in this party. It got me thinking, what exactly is going on inside that tank? What are these elements, best known for their distinct glowing personalities, doing all mixed up together?
It’s like having a celebrity party in a confined space. You’ve got the bubbly, buoyant Helium, always ready for a good time (and a high-pitched voice). Then there’s the charismatic Neon, the life of the party, practically begging to be plugged into an electrical current to show off. And finally, Argon, the quiet observer, the one who’s just… there. It’s an odd bunch, really. What’s the point of them hanging out together in a tank? Are they plotting something? A gas rebellion?
This isn’t just a random question, you know. Understanding mixtures like this is actually pretty fundamental to a whole bunch of things we see and use every day. It’s not just for mad scientists in their dusty corners. Think about it, even if you’re not building a particle accelerator in your garage (though, hey, no judgment if you are!), the principles behind how these gases interact, or rather don't interact, are fascinating.
The Noble Gang: A Reputation Precedes Them
Let’s talk about these guys individually for a sec. Helium (He), Neon (Ne), and Argon (Ar). They’re all part of this special club in the periodic table called the noble gases. And let me tell you, they live up to their name. They’re noble because they’re incredibly… well, unreactive. Imagine the most introverted, socially awkward people you know, but on an atomic level. That’s pretty much them.
Why are they so aloof? It all comes down to their electron shells. Atoms, as you probably remember from high school (or maybe you blissfully blocked it out, I can relate), have electrons whizzing around the nucleus. These electrons are arranged in shells or energy levels. The outer shell, the one that interacts with other atoms, is the key player. Noble gases have a full outer electron shell. It’s like they’ve achieved atomic enlightenment. They’ve got everything they need, and they’re not looking to gain, lose, or share any electrons. They’re perfectly content in their stable, electron-filled universe.
This inherent stability is what makes them so useful. Because they don’t want to react with anything, they can be used in environments where reactivity would be a disaster. Think about welding, or those fancy light bulbs that last for ages. Yup, you guessed it, noble gases are often involved.

Helium: The Light and Airy One
Helium, number two on the periodic table, is the lightest of the noble gases. It’s famous for two things: making balloons float and giving you that squeaky voice when you inhale it. Please tell me you’ve done the squeaky voice thing. It’s a rite of passage! Its low density compared to air is why it’s used for balloons and airships (though for safety, it's more common than the highly flammable hydrogen now).
But Helium’s noble status is crucial here. It’s not going to react with the rubber of the balloon, or the material of the airship. It just sits there, being light and unreactive. It’s also used in cryogenics, for cooling superconducting magnets in things like MRI machines. Imagine trying to cool something down with a gas that’s busy forming chemical bonds with everything it touches. Not ideal, right?
Neon: The Star of the Show
Neon, with its atomic number of ten, is perhaps the most visually striking of the bunch. When you pass an electric current through pure Neon gas, it emits a beautiful, bright reddish-orange light. That’s the classic neon sign glow. It’s incredibly vibrant and eye-catching. Back in the day, it was the go-to for signage because it was so distinctive and, well, cool.
But Neon isn’t just about glowing. It’s also used in plasma televisions and as a refrigerant in some specialized applications. Again, its inert nature is paramount. It doesn’t degrade the components it’s used with, ensuring longevity and performance. It’s the diva of the noble gases, but a reliable one.
Argon: The Understated Workhorse
And then there's Argon. Atomic number 18. It’s the most abundant noble gas in Earth's atmosphere, making up about 0.93%. It’s the quiet one, the one you might not think much about, but it’s actually incredibly important. Argon is colorless, odorless, and tasteless, and its main claim to fame is its excellent protective properties.

Think about welding. When you’re welding metal, you need to prevent the molten metal from reacting with the oxygen and nitrogen in the air, which would weaken the weld and cause defects. Argon is often used as a shielding gas for this reason. It creates an inert atmosphere around the weld, protecting it. It’s like a silent bodyguard for your molten metal.
Argon is also used in incandescent light bulbs. In older bulbs, a vacuum was used, but this could cause the filament to evaporate more quickly. Filling the bulb with an inert gas like Argon (or a mixture including Nitrogen) slows down the evaporation of the filament, making the bulb last longer. It’s a subtle but significant contribution to everyday life.
The Cocktail Hour: Why Mix Them?
So, if these gases are so happy doing their own thing, why would anyone mix them? What’s the point of this particular blend of Helium, Neon, and Argon? It’s not about creating some super-gas with enhanced reactivity – that’s the exact opposite of what noble gases are good for!
The answer, as it often is in science, is about fine-tuning. Different mixtures can produce different results, especially when you introduce energy, like electricity. Each gas, when excited, emits light at specific wavelengths, giving it a characteristic color. When you mix them, you’re not creating a new chemical substance, but rather a new luminescent blend.
Imagine you’re a painter and you’ve got three primary colors. Red (Neon), a bit of blue (let’s pretend Helium can do blue for a second, it doesn’t really, but work with me here), and maybe a neutral grey (Argon, being pretty much invisible). By mixing these, you can create a whole spectrum of new colors. It’s similar, but instead of pigments, you have different gases that emit light.

Specialized Lighting: Beyond the Basic Glow
One of the most common reasons for mixing Helium, Neon, and Argon is for specialized lighting applications. While pure Neon gives you that classic red, mixing it with other gases can produce a wide range of colors. For instance:
- Neon + Argon: This combination can produce blues and greens. Argon, while not emitting a strong visible light on its own in the same way Neon does, can modify the electrical discharge and influence the wavelengths of light emitted by Neon.
- Adding Helium: Helium can be used to alter the electrical characteristics of the gas discharge. It can also contribute to the overall luminescence. While Helium itself doesn't have a prominent visible emission in this context, its presence can affect how the other gases behave.
This is how you get those amazing, multi-colored displays. Think about those intricate gas-discharge tubes used in art installations, high-end signage, or even in some scientific instruments. The exact ratio of Helium, Neon, and Argon is carefully controlled to achieve the desired color, brightness, and operational characteristics.
It’s a bit like a chef creating a complex dish. You have your core ingredients, but it’s the precise proportions and the way you cook them that create the final flavor. In this case, the "cooking" is the electrical current, and the "flavor" is the light produced.
Industrial Applications: More Than Just Pretty Lights
Beyond aesthetics, these mixtures can also have practical industrial uses. In certain gas lasers, mixtures of noble gases are used to generate coherent light. The specific combination of gases and the way they are excited determines the wavelength and power of the laser beam.
For example, the Helium-Neon (HeNe) laser is a very common type of laser. It's known for its stable output and its characteristic red beam. It works by exciting a mixture of Helium and Neon. The Helium atoms are excited first, and then they transfer their energy to the Neon atoms, which then emit photons of light. Argon can also be part of more complex gas laser mixtures for different wavelengths.

This is pretty mind-blowing when you think about it. These gases, so inert on their own, become the very engine of precise, high-energy light beams that are used in everything from barcode scanners to surgical tools. It’s a testament to how understanding the fundamental properties of matter can lead to incredibly advanced technologies.
The Chemistry of "No Chemistry"
What's so fascinating about these noble gas mixtures is that the lack of chemical reaction is the whole point. We’re not trying to make them bond. We’re trying to exploit their individual properties when they’re forced into a situation where they might want to react, but they won’t. It’s like sending a highly trained, disciplined army into a chaotic battle – they stand firm, perform their designated tasks, and don’t get bogged down by the surrounding mayhem.
The tank in my friend’s apartment, with its seemingly random assortment of inert gases, is essentially a reservoir of potential. It holds the building blocks for a controlled release of energy in the form of light. It’s a silent promise of a specific glow, a predictable discharge, a reliable shielding. It’s the chemistry of non-chemistry, the science of stable entities collaborating through their very refusal to engage.
So next time you see a colorful neon sign, or use a laser pointer, or even just marvel at a helium balloon bobbing away, remember that there’s a whole lot more going on than meets the eye. There are these noble gases, minding their own business, full outer shells intact, but when we coax them with a little bit of energy, they can put on quite a show. And sometimes, they do it together, a perfectly orchestrated dance of inertness, creating the very spectrum of light we perceive.
It makes you wonder what other seemingly simple mixtures hold hidden depths, doesn't it? The world is full of these quiet collaborations, these unseen interactions. And it all starts with understanding the fundamental building blocks, even the ones that are too proud to join the party. Kinda makes you feel a bit sorry for them, doesn't it? All that potential for interaction, just… unfulfilled.
