A Student Was Titrating A Solution Of Hydrazine

So, get this. A student was doing something super nerdy. Like, really nerdy. They were titrating a solution of hydrazine. Woah, right?
Okay, okay, I know what you’re thinking. “Hydrazine? Titrating? Sounds like my worst nightmare.” But stick with me! It’s actually kinda cool. And a little bit dangerous. Which, let’s be honest, makes it way more interesting.
What in the World is Hydrazine?
First off, hydrazine. What even is it? It’s this weird chemical compound. Think of it as Nitrogen’s edgy cousin. Its formula is N₂H₄. Two nitrogen atoms, four hydrogen atoms. Simple, right? But don't let the simplicity fool you.
This stuff is reactive. Like, super reactive. It’s a powerful reducing agent. That’s science-speak for it likes to give away electrons. And when it gives away electrons, things happen. Exciting things. Sometimes explosive things.
Historically, hydrazine had a bit of a reputation. It was used in rocket fuel. Yeah, rocket fuel! Like the stuff that blasts things into space. So, this student is playing with something that could potentially launch them into orbit. Or at least make a very impressive pop.
It’s also a known carcinogen. So, you know, wear your safety goggles and a hazmat suit. Just kidding! (Mostly.) In a lab setting, with proper precautions, it’s manageable. But it’s good to know you’re dealing with something a little bit… feisty.
Titrating: The Science of Measuring
Now, what’s this “titrating” business? Imagine you have a mystery drink. You want to know how much sugar is in it. Titration is like a super precise way of figuring that out.
You take a known amount of your mystery drink. Then, you slowly add another liquid – called a titrant – until something specific happens. Like, maybe the drink changes color. Or a reaction is complete.
The titrant has a known concentration. So, by seeing how much of the titrant you need to add to get that specific result, you can calculate the concentration of your original mystery drink. It’s like a chemical detective story!
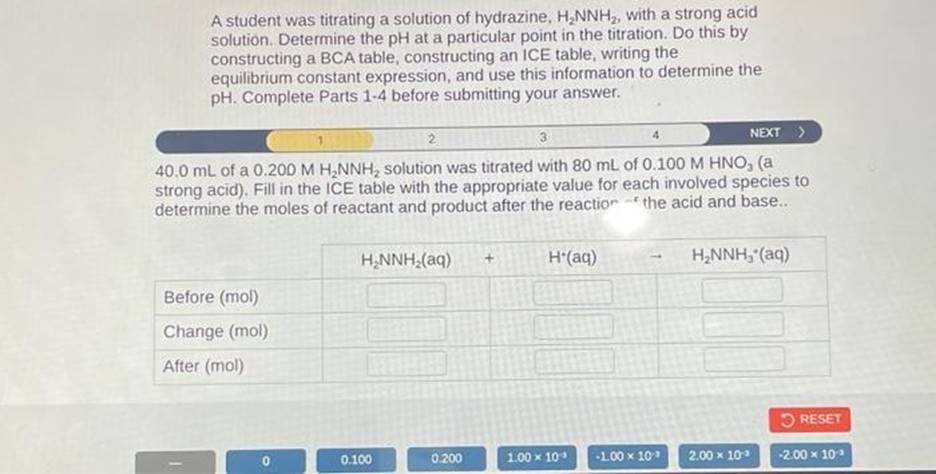
In our case, the student is titrating hydrazine. They’re probably using it to determine the concentration of another solution. Or maybe they’re seeing how much of something else hydrazine can react with. It’s all about precision and control. Which is, in itself, a fun challenge.
Why is Hydrazine Titration So Cool?
Okay, so we have a reactive, potentially explosive chemical, and a precise measuring technique. What’s the fun in that? Oh, let me count the ways!
Firstly, the stakes. When you’re titrating something as notoriously energetic as hydrazine, every drop matters. You can’t just slosh it in there. You’ve got to be deliberate. It’s like defusing a tiny, liquid bomb. With science!
Secondly, the visual. While some titrations are about subtle color changes, hydrazine reactions can be… more dramatic. Think of unexpected fizzes. Or maybe even a little smoke. Nothing too crazy, hopefully, but enough to make your heart skip a beat (in a good way, of course).
Thirdly, the historical context. This isn’t just some random chemical. Hydrazine powered the very first rockets that took humans to the moon! It’s part of the history of exploration and innovation. So, when a student is working with it, they’re tapping into a legacy of pushing boundaries.
And let’s not forget the element of surprise. Chemistry, especially with reactive compounds, can always throw a curveball. A slight impurity, a change in temperature, a tiny bit too much titrant… suddenly, you have an unexpected result. Which, for a scientist, is often the most exciting part!
The Quirky Details
Did you know hydrazine is also used in pharmaceuticals? Yep. Some drugs use it as a building block. So, it’s not just for rockets and explosions. It’s also for making you feel better. Kind of a dual personality, wouldn’t you say?
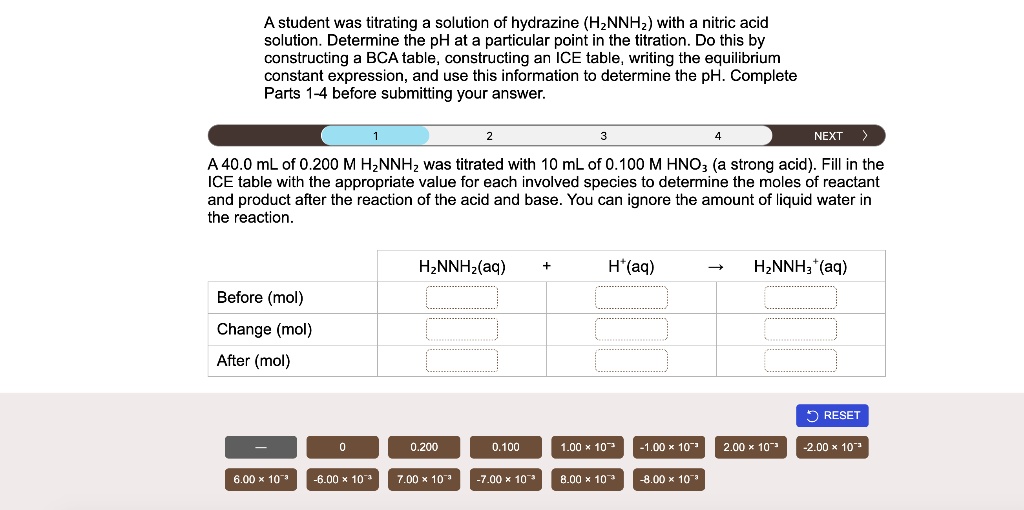
And here’s a fun fact: pure hydrazine is unstable. It can decompose explosively. So, it’s often handled in diluted solutions. Still potent, but a little less likely to say “boom!” at an inconvenient moment.
The smell of hydrazine is often described as ammonia-like. So, imagine the lab smelling like a very strong cleaning product. Not exactly Chanel No. 5, but it’s the smell of science in action!
Titration itself is an art form. It requires patience, steady hands, and a keen eye. When you’re dealing with hydrazine, those skills are amplified. You’re not just measuring; you’re performing a delicate dance with a powerful substance.
The Student's Adventure
So, this student, armed with their beakers, burettes, and a healthy dose of caution, is carefully adding their titrant to the hydrazine solution. They’re watching for that tell-tale sign. Maybe a color change from clear to a vibrant blue. Or perhaps a sudden effervescence.
Every milliliter of titrant is a victory. Every observation is a clue. They’re learning not just about chemistry, but about the properties of matter, the thrill of discovery, and the importance of meticulous work.
It’s a moment of intense focus. The rest of the world fades away. It’s just the student, the chemicals, and the silent, unfolding reaction. It’s pure, unadulterated science. And that, my friends, is pretty darn cool.
So, next time you hear about someone titrating hydrazine, don’t just think “boring lab work.” Think of rockets. Think of chemical detectives. Think of the brave student carefully maneuvering a substance that powers our ambitions and pushes the boundaries of what’s possible. It’s a little bit wild, a little bit dangerous, and a whole lot of fun to think about.
