A Student Performs An Acid-base Titration And Plots

Hey there, ever feel like you’re just going through the motions sometimes? Like, you’re doing something, but you’re not quite sure why it matters? Well, buckle up, buttercup, because we’re about to dive into the wonderfully weird world of acid-base titrations, and I promise you, it’s way cooler and more relevant than it sounds. Think of our intrepid student, let’s call her Maya, carefully pouring liquids and scribbling notes. She’s not just mixing chemicals for the fun of it; she’s on a mission to uncover some hidden truths. And guess what? The principles she's playing with are popping up all around you, every single day.
So, what exactly is this “titration” thing? Imagine you’re trying to figure out just how much sugar is in your grandma’s famous lemon pie. You can’t just eyeball it, right? You need a way to measure it. A titration is kind of like that, but with acids and bases. Acids are those tangy, sour things (think lemon juice, vinegar, or that fizzy sensation from a soda). Bases are their opposite; they’re often bitter and feel a bit slippery (like baking soda paste, or even some soaps).
Now, acids and bases are like frenemies. They really like to react with each other, and when they do, they neutralize each other. It’s like the ultimate compromise: a strong opinionated acid meets a strong opinionated base, and poof! They become pretty much neutral. A bit like when your super energetic toddler finally calms down after a good nap, or when your grumpy cat decides to tolerate a head scratch. Harmony achieved!
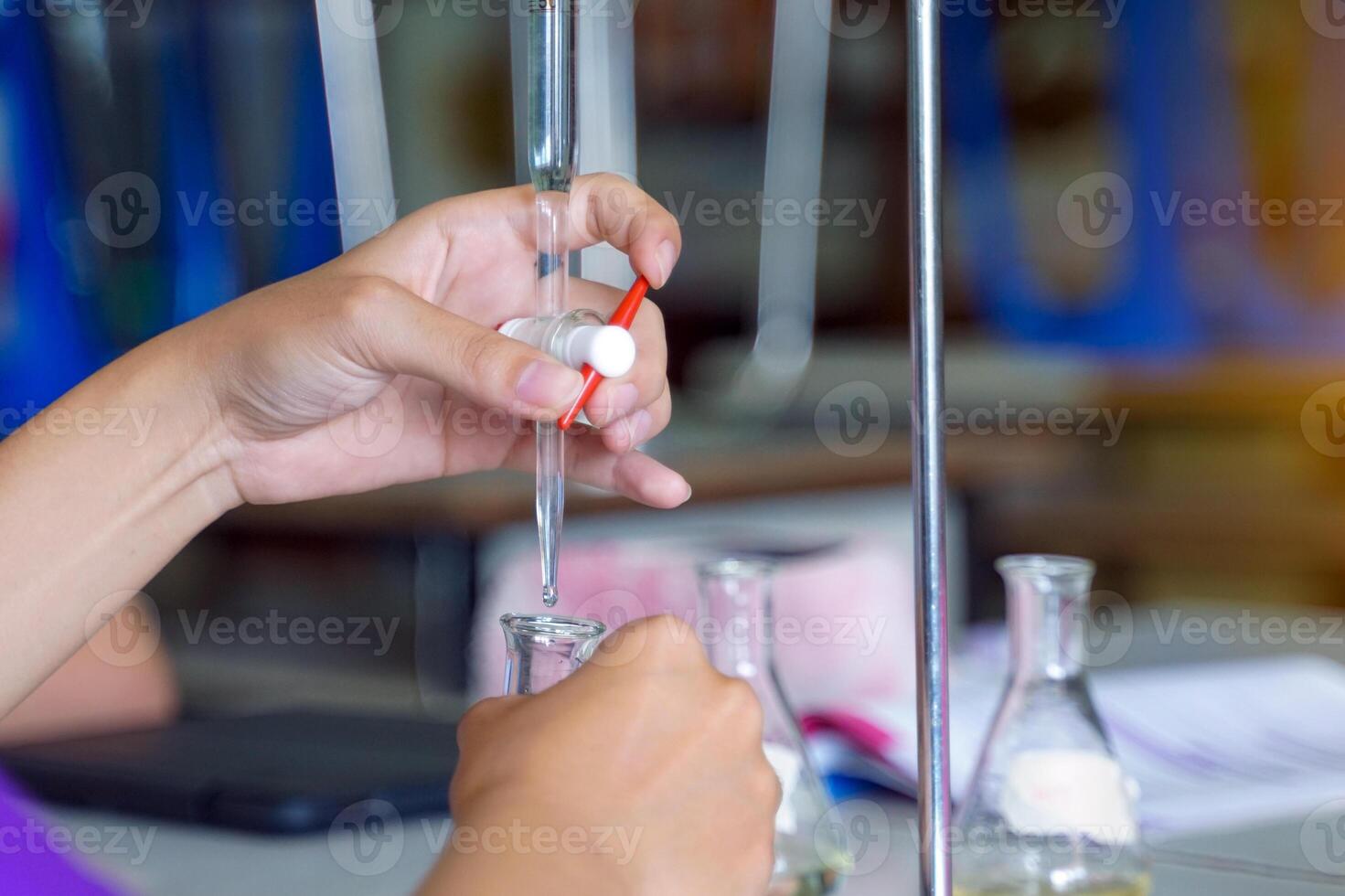
Maya’s titration involves taking a known amount of one substance – say, a mystery acid in a flask – and then slowly adding a solution of the other, a base, from a fancy-looking dropper called a burette. She’s adding it drop by tiny drop, like a chef carefully seasoning a dish, making sure not to overdo it. The goal is to find the exact point where the acid and base have perfectly canceled each other out. This magical moment is called the equivalence point.
How does she know she’s reached this magical point? Well, scientists are clever cookies. They use something called an indicator. Think of an indicator as a mood ring for your solution. It’s a special chemical that changes color when the balance shifts just right. For example, phenolphthalein (try saying that five times fast!) is clear in acidic solutions but turns a vibrant pink in basic solutions. So, Maya watches her solution like a hawk. The moment it just starts to blush pink, she stops adding the base. Ta-da! She’s found her equivalence point.

But why should you care about Maya’s pink-tinged beakers? Because this seemingly simple act of mixing and observing has some seriously everyday implications. Let’s talk about your stomach. You know that burning feeling, that heartburn? That’s usually stomach acid being a bit too enthusiastic. And what do you often reach for? An antacid! Antacids are essentially bases. They’re designed to neutralize that excess stomach acid, bringing your digestive system back to a happy, neutral pH. So, when you pop an antacid, you’re essentially performing a mini acid-base reaction in your own body, just like Maya, only a lot less precise and probably less pink!
Think about baking, too. Baking soda (a base) reacts with acidic ingredients like buttermilk or lemon juice to create those lovely bubbles that make cakes and cookies light and fluffy. Get the ratio wrong, and your cake might be flatter than a pancake after a steamroller convention. Understanding these reactions, these titrations of sorts, is crucial for getting those delicious results. It’s all about finding that perfect balance, that sweet spot where everything just works.
Maya doesn't just stop at finding the equivalence point, though. She takes her data – how much base she added, the concentration of her solutions – and then she does something really cool: she plots it. She creates a graph, a visual story of her experiment. This graph, often called a titration curve, shows how the pH of the solution changes as the base is added. It’s like a temperature chart for a patient’s recovery, or a stock market graph showing trends, but for chemical reactions.
These graphs are super informative. They can tell us not just how much acid or base is present, but also give us clues about the strength of the acid or base. A steeper curve might indicate a stronger acid or base, while a gentler slope suggests a weaker one. It’s like looking at a report card for Maya’s chemicals – she can see their performance and understand their character better.

This concept of plotting and analyzing data is huge. It’s how scientists learn about everything from the medicines we take to the quality of the water we drink. Water treatment plants, for instance, regularly test the pH of water. They might perform titrations to ensure the water is not too acidic or too alkaline, which could be harmful to us and to the environment. Imagine if the chemicals used to clean your drinking water were out of whack – yikes! So, Maya’s graphing isn’t just for a grade; it’s about understanding and controlling the world around us.
Even something as simple as cleaning can involve acid-base chemistry. Many household cleaners are either acidic or basic. Bleach, for example, is a base. Vinegar is an acid. When you mix them carelessly, you can create some pretty nasty fumes! It’s a stark reminder that understanding these reactions, and knowing when to add what, is super important for safety. Maya’s meticulous work is a much safer way to explore these interactions.
So, next time you reach for that antacid, or enjoy a perfectly fluffy muffin, or even just wonder about the cleanliness of your tap water, remember Maya and her titration. Remember that behind those everyday conveniences and wonders, there’s often a careful dance of acids and bases, a quest for balance, and the powerful insights gained from a simple plot. It’s a reminder that even in a lab coat, we’re all just trying to figure things out, one drop, one data point, one satisfyingly neutral solution at a time. And that, my friends, is pretty darn cool.
