A Species Whose Oxidation Number Increases In A Reaction Is
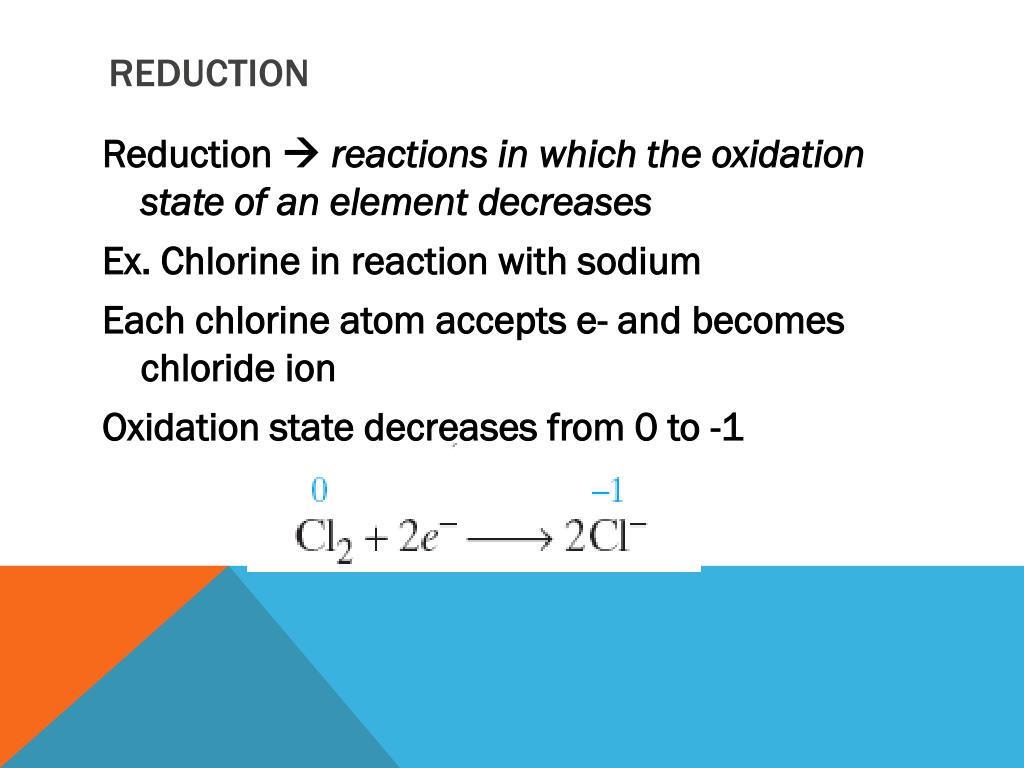
Ever wondered what's happening behind the scenes when you see a rusty bike or a colorful fireworks display? It's all thanks to some super cool chemistry, and today we're diving into a particularly fascinating aspect: a species whose oxidation number increases in a reaction. Now, that might sound a bit technical, but stick with me, because understanding this is like unlocking a secret code to how a lot of everyday magic happens! It’s fun because it explains things we see all the time, and incredibly useful for understanding everything from cooking to how our bodies work.
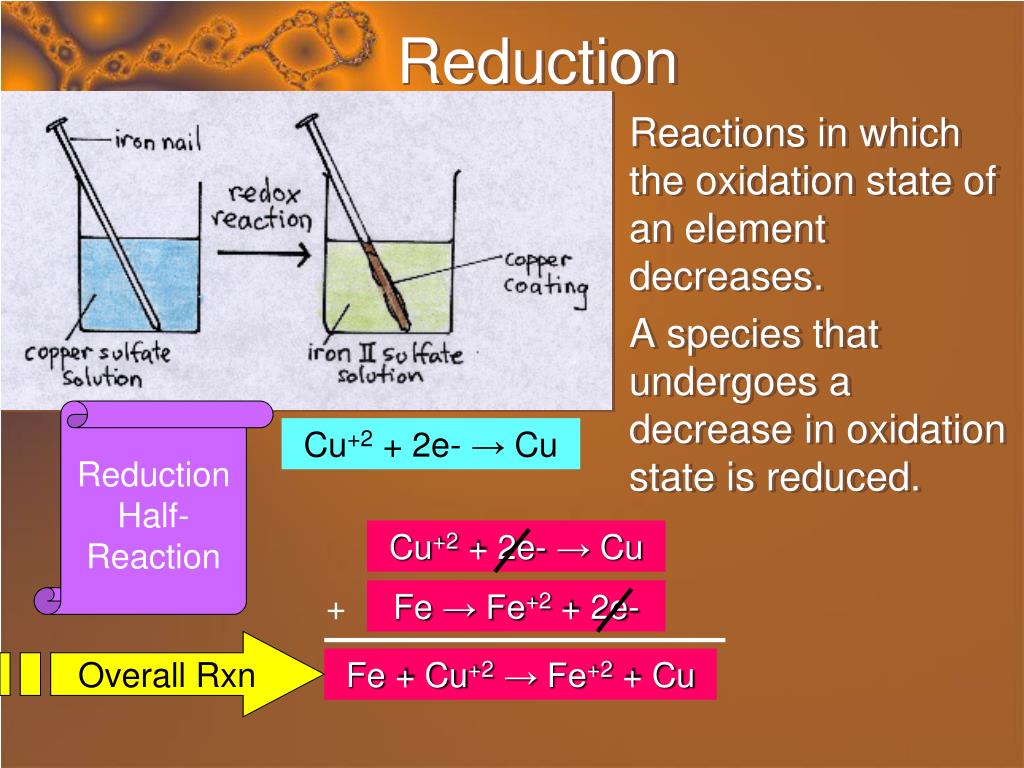
So, what exactly are we talking about? Think of oxidation numbers as a way to keep track of electrons in a chemical reaction. When an atom or molecule's oxidation number increases, it means it's losing electrons. This process is called oxidation. It’s like a chemical high-five, where one species gives away electrons and another accepts them.
Why is this useful? For beginners, it’s a fundamental building block for understanding chemistry. It helps explain why certain reactions happen and others don't. For families, it can be a fun way to explore concepts with kids. Think about the browning of an apple – that’s oxidation in action! For hobbyists, whether you're into metalworking, brewing, or even gardening, understanding oxidation can help you achieve better results and prevent unwanted changes.
Let's look at some simple examples. The most common one is rusting. When iron (Fe) reacts with oxygen (O₂) in the air and water, the iron's oxidation number goes from 0 to +3, forming iron oxide (rust). The iron is being oxidized. Another classic is the reaction of a metal like magnesium (Mg) with oxygen to form magnesium oxide (MgO). Magnesium starts with an oxidation number of 0 and ends up with +2. It's been oxidized!
Even things like breathing involve oxidation. When we digest food, complex molecules are broken down, releasing energy. This is a biological form of oxidation where our cells effectively "burn" fuel for energy, and certain atoms within those molecules see their oxidation numbers increase.
Ready to get started? You don't need a fancy lab. Observe! Look around your home. See that tarnished silver? That's oxidation. Notice how batteries work? They rely on controlled oxidation-reduction (redox) reactions. You can even do simple experiments with common household items. For instance, try observing how a cut piece of potato turns brown when exposed to air – that’s an enzyme-catalyzed oxidation.
Understanding that a species whose oxidation number increases is undergoing oxidation is a gateway to a whole world of chemical understanding. It’s not just about equations; it’s about the dynamic interactions that shape our world, from the grand scale of planetary atmospheres to the intricate processes within our own bodies. Embracing these basic chemical concepts can make everyday observations even more fascinating and rewarding.
