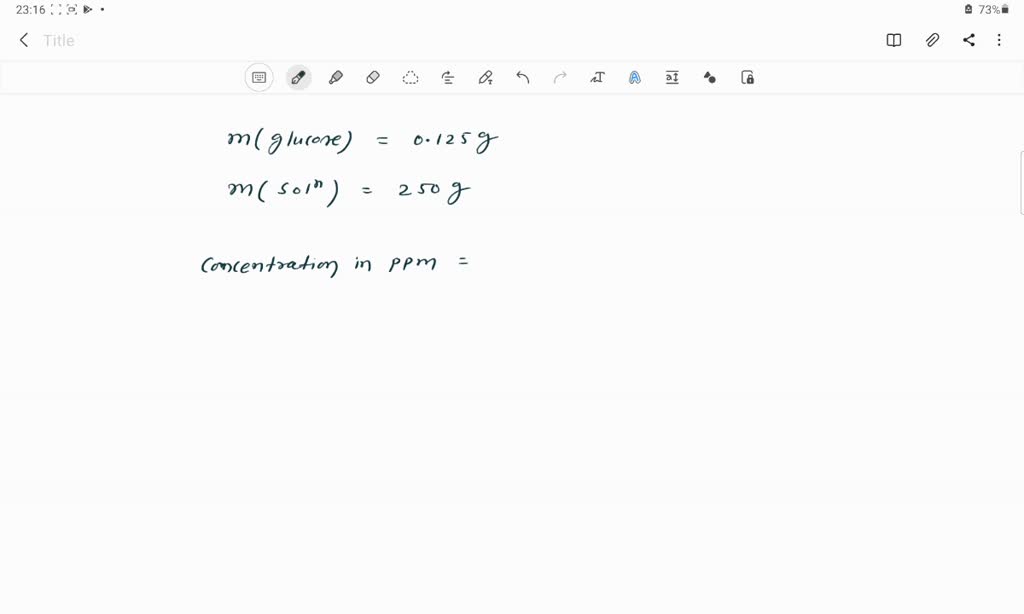
A Solution Is Prepared Using 0.125 G Of Glucose
Hey there, you wonderful humans! Ever stopped to think about the tiny, almost invisible things that make our world tick? We’re talking about the stuff that powers our bodies, fuels our thoughts, and, yes, even makes a perfectly brewed cup of tea taste just right. Today, we’re diving headfirst into a world of minuscule measurements and super important ingredients. Get ready, because we’re about to uncover the magic hidden in a mere 0.125 grams of glucose. Yep, you read that right!
Now, I know what you might be thinking. “Glucose? Isn’t that just… sugar?” Well, yes and no! While glucose is indeed a type of sugar, it’s also so much more. Think of it as the ultimate energy currency for your cells. It’s the primary fuel source that keeps everything from your brain firing on all cylinders to your muscles flexing like a superhero. And the amount we’re talking about today? 0.125 grams? It might sound like pocket change, but let me tell you, it’s a pretty big deal when you break it down.
The Tiny Titan of Energy
So, how much is 0.125 grams, anyway? Let’s get visual. Imagine a small pinch of sugar. Like, really small. Maybe a few individual crystals. That’s roughly it! It’s so little that if you were to measure it out casually, you might miss it entirely. But in the grand scheme of things, this minuscule amount is packed with potential. It’s like a tiny seed that holds the blueprint for a mighty oak tree – all the power is there, just waiting to be unlocked.
When we talk about solutions, especially in the realm of science and even just in our kitchens, we’re usually mixing something solid into a liquid. And when that something is glucose, even this tiny amount can have some pretty fascinating applications. It’s all about concentration, you see. The ratio of glucose to the liquid is what determines its effectiveness, and even a whisper of glucose can make a significant difference.
Fueling the Everyday Hero: YOU!
Let’s bring it back to you, the incredible person reading this. Your body is an amazing machine, and it’s constantly working to keep you going. Ever feel that mid-afternoon slump? That’s often your body signaling that it’s running a little low on glucose. That tiny pinch of 0.125 grams, if it were part of a larger, perfectly balanced intake, would be contributing to your brain’s ability to focus, your heart’s steady beat, and your overall vitality. It’s the unsung hero of your daily energy levels!
Think about it: that feeling of alertness after a good meal, the energy you have for your hobbies, the sheer joy of being able to run around with your kids or pets. All of that, in part, is thanks to the glucose your body efficiently processes. And who’s to say that even a tiny, precisely measured amount isn’t playing its crucial role in that complex symphony of life?
Beyond the Body: Sweet Science in Action
But the story of 0.125 grams of glucose doesn’t end with our personal physiology. This humble ingredient pops up in some really cool places in the world of science and technology. For instance, in laboratories, scientists often prepare very specific solutions for experiments. These solutions might be used to grow delicate cell cultures, to test the efficacy of new medicines, or even to study the intricate workings of biological processes.
And guess what? Sometimes, the perfect recipe calls for just a dash of glucose. That 0.125 grams could be the precise amount needed to provide just enough food for a microscopic organism to thrive, or to create a medium that mimics the conditions found inside a living cell. It’s all about precision and understanding the power of even the smallest quantities. It’s like being a master chef, but instead of salt and pepper, you’re working with fundamental building blocks of life!

A Touch of Sweetness for the Modern World
Even in our everyday lives, the concept of solutions involving small amounts of glucose can be surprisingly relevant. Think about certain sports drinks or electrolyte beverages. While they contain more than just glucose, the inclusion of simple sugars like glucose is essential for rapid energy replenishment. The careful formulation ensures you get the boost you need without overwhelming your system. And at the heart of that formulation, at some stage, a precise amount of glucose was likely measured and dissolved.
Furthermore, in the world of baking, while we often use larger quantities, the principle remains the same. Glucose, often in a syrup form, can affect the texture and shelf-life of baked goods. Even a tiny adjustment can lead to a wonderfully chewy cookie or a perfectly risen cake. It’s the little things, the details, that make all the difference, wouldn’t you agree?

Making Life More Fun (Seriously!)
Okay, okay, I can see you raising an eyebrow. “How can 0.125 grams of glucose make my life more fun?” Bear with me! It’s about appreciating the underlying science that makes so many enjoyable things possible. Understanding that this tiny amount is a key player in fueling your adventures, from a challenging hike to a lazy afternoon reading your favorite book, adds a layer of appreciation, doesn't it?
Imagine the joy of learning about how your body works, how food transforms into energy, and how scientists are using these fundamental components to solve complex problems. It’s like unlocking a secret level in a video game, where you gain a deeper understanding of the world around you. And honestly, that’s pretty darn fun!
Plus, think about the culinary arts! When you bake that perfect loaf of bread or whip up a batch of cookies, you’re essentially creating a solution where ingredients, including sugars like glucose, interact in fascinating ways. Understanding the science behind it can elevate your cooking and baking from simply following a recipe to becoming a true culinary artist. You can experiment, understand why things happen, and create even more delicious outcomes. That’s a win-win in my book!

The Inspiring Power of Tiny Contributions
So, the next time you see a measurement, no matter how small, remember the incredible potential it holds. That 0.125 grams of glucose isn’t just a number; it’s a spark of energy, a building block of life, and a testament to the intricate beauty of chemistry and biology. It’s a reminder that even the smallest components can have a profound impact.
It’s about appreciating the precision, the science, and the sheer ingenuity that goes into creating the world we live in. From the food we eat to the medicines that heal us, these tiny, carefully measured ingredients are the silent architects of our well-being and our enjoyment. They’re proof that big things often come in small packages.
Doesn’t that just make you want to explore more? To ask questions? To marvel at the hidden wonders all around us? The world is full of these amazing, often unseen, processes. So, go ahead, be curious! Delve into the fascinating world of science, nutrition, and everyday chemistry. You might be surprised at how much fun you can have discovering the magic in the minuscule. The adventure of learning is always in season, and with a little curiosity, you’ll find inspiration everywhere you look!
