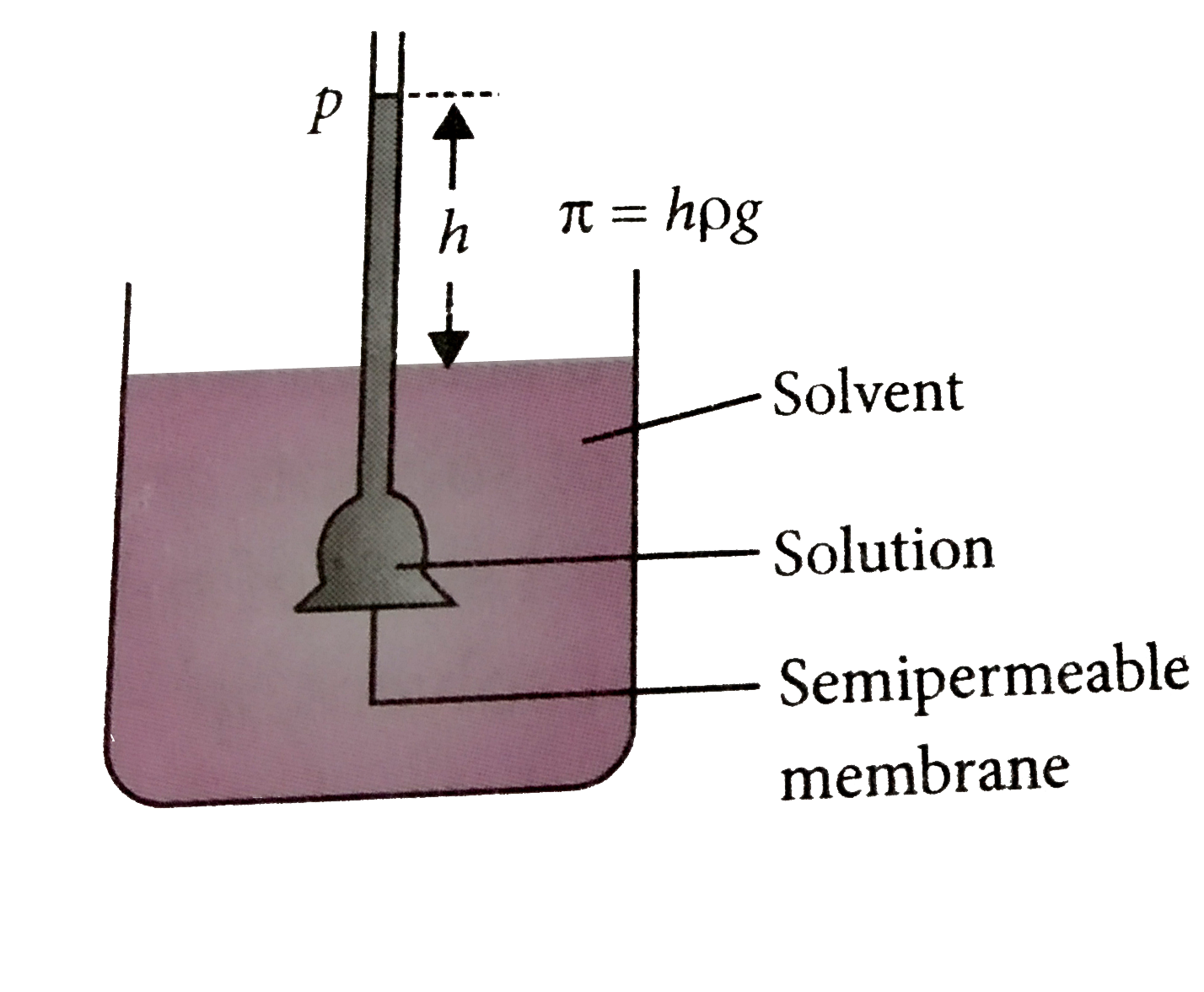
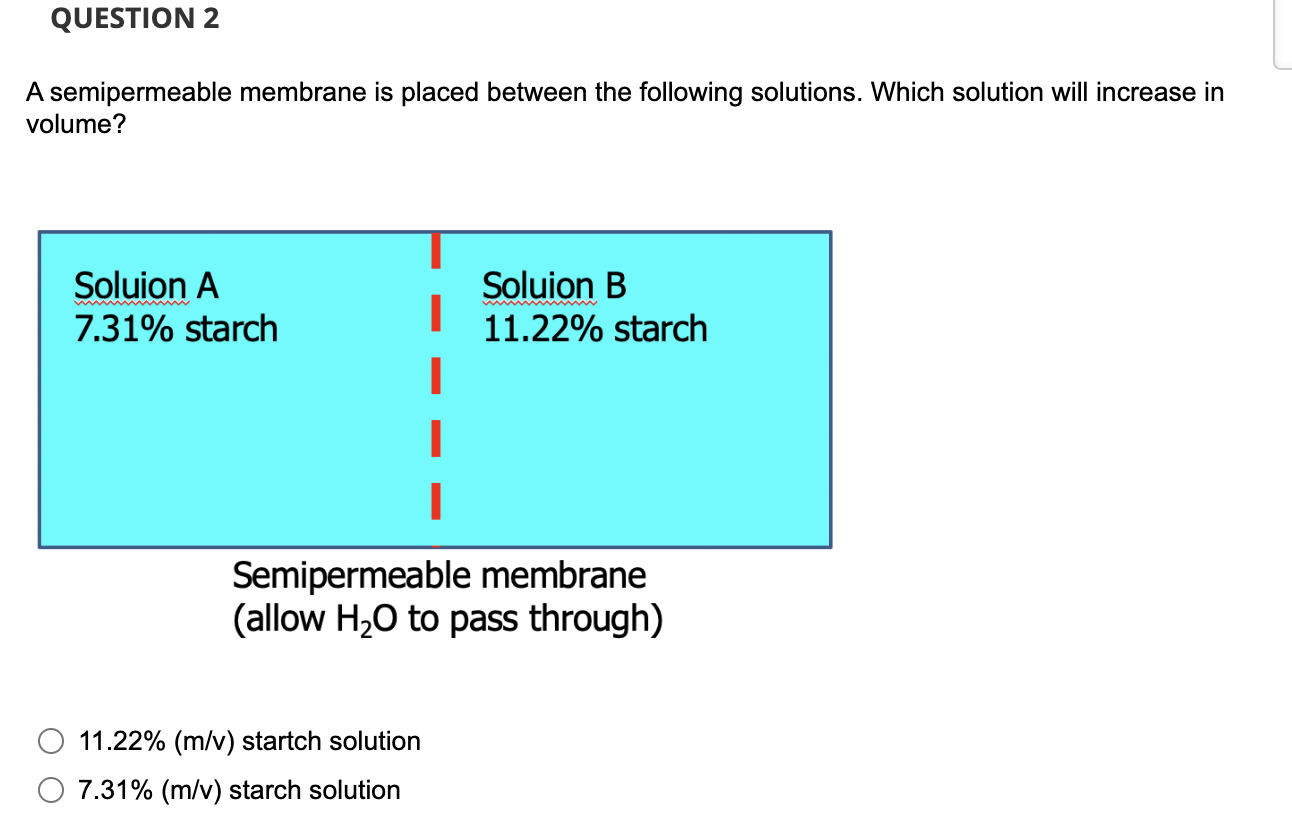
A Semipermeable Membrane Is Placed Between The Following Solutions
Ever feel like your kitchen is a tiny, bustling microcosm of the universe? Well, you're not entirely wrong! Today, we're going to dive into something called a "semipermeable membrane," which sounds like it belongs in a sci-fi movie or a very intense chemistry lab, but trust me, it’s something you’ve totally encountered in your own life. Think of it as the ultimate picky bouncer at the club of your cells, or maybe that one friend who only lets you borrow their favorite sweater, but never their car.
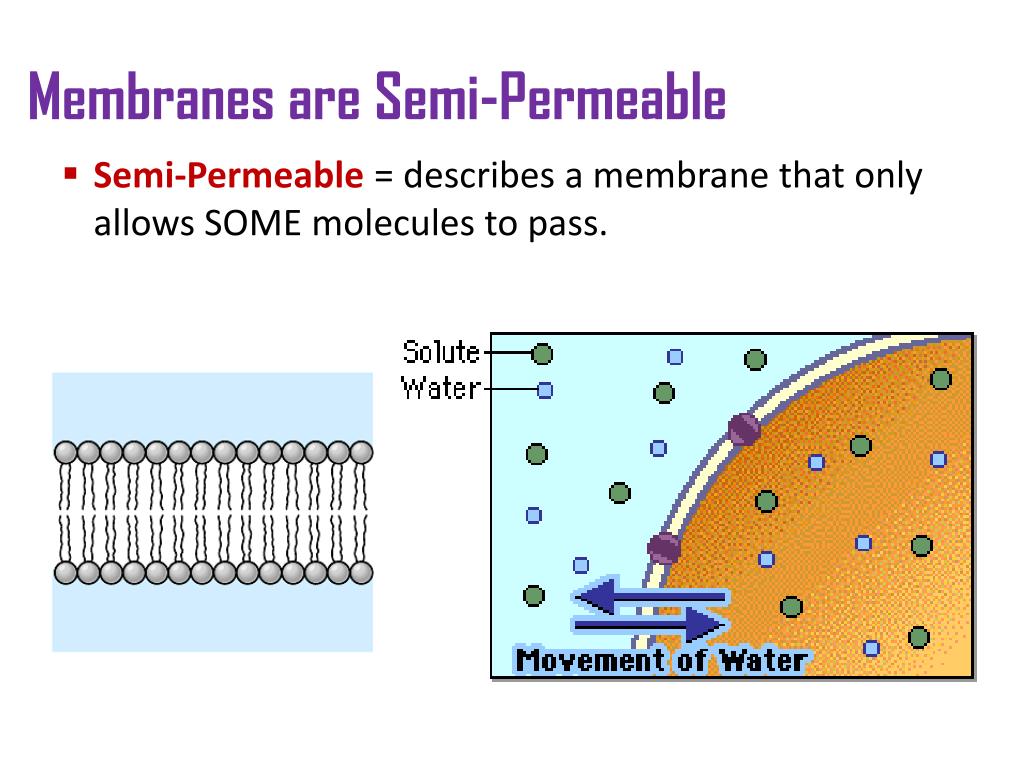
So, what's the deal with this fancy-sounding membrane? Basically, it’s a barrier that’s a bit of a selective shopper. It lets some things through, like a chill friend handing out free samples, but it puts up a firm "nope" to others, like that nagging voice in your head telling you not to eat the entire bag of chips. It’s all about controlling the flow, deciding who gets the VIP pass and who has to wait outside.
Imagine you've made a super-salty batch of popcorn (because who doesn't love salty popcorn?) and your friend, who's only drinking plain water, looks a bit parched. Now, if you were to put a special, selective barrier between your popcorn bowl and their water glass – that, my friends, is our semipermeable membrane in action. It's like a gatekeeper saying, "Okay, little water molecules, you can waltz on through. But you, Mr. Salt, you're too big for this party. Stay right there."
This whole concept is actually the secret sauce behind a lot of things we take for granted. Think about how your body keeps all its important bits and bobs exactly where they need to be. Your cells are basically little houses, and each cell has its own semipermeable membrane – its front door and its security system. It lets in the good stuff, like nutrients and water, which are like the pizza delivery and the comfy couch, essential for a good time. But it keeps out the bad stuff, like germs and waste products, which are like that awkward relative who overstays their welcome or the dodgy spam email.
Let’s get a little more specific, shall we? We’re going to set up a little scenario, a bit like staging a miniature science experiment in your mind. We're going to take two solutions, which are just fancy words for things dissolved in water. Think of them as two different kinds of Kool-Aid, maybe one super sweet cherry and the other a rather bland lime.
Now, we plop our trusty semipermeable membrane right in the middle, like a referee stepping onto a slightly sticky sports field. On one side, we have Solution A. Let's make Solution A a nice, concentrated sugary drink. Imagine it’s that time of day when you desperately need a sugar rush, and this is your ultimate solution. We're talking lots of sugar molecules, practically a mosh pit of sweetness.
On the other side, we have Solution B. For Solution B, we’re going to go with something much less exciting. Let’s say it’s just plain water. Not even sparkling water, just good old H₂O, minding its own business. So, on one side, you have a veritable buffet of sugar, and on the other, you have a quiet, humble glass of water.
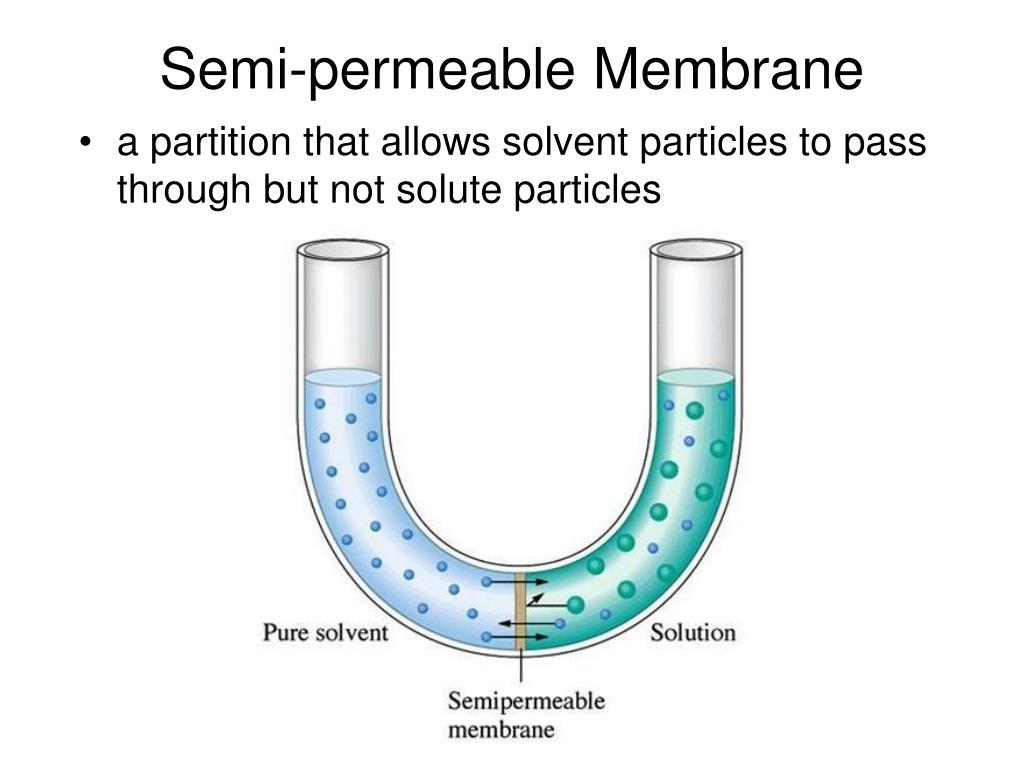
The semipermeable membrane, our selective friend, is now faced with this situation. What does it do? Well, it’s got its rules, you see. The membrane has tiny little holes, like a colander, but even tinier. These holes are just the right size for water molecules to sneak through. They’re like the small, agile dancers at a party, able to weave through the crowd easily. However, those big, bulky sugar molecules in Solution A? They’re more like the awkward uncle trying to do the worm – they’re just too large to fit through the tiny openings in the membrane.
So, what happens next? Nature, being the incredibly clever and sometimes slightly bossy force that it is, kicks in. There’s a natural tendency for things to try and balance out. Think of it like a seesaw. If one side is way heavier, the lighter side wants to join the party to even things out. In our case, the water molecules on the side with less "stuff" (Solution B, our plain water) are going to start feeling a bit lonely. They see all the action happening on the sugar side, and they think, "Hey, why not join the fun?"
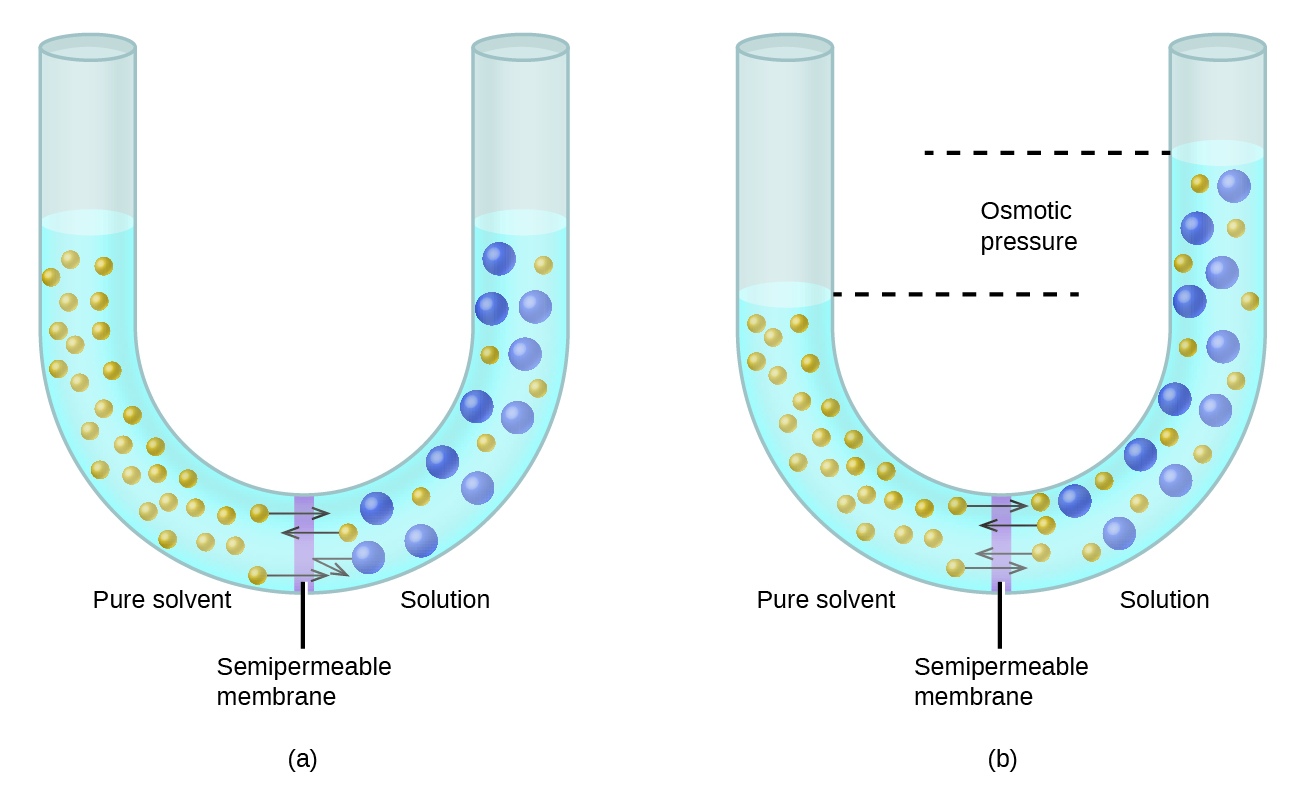
So, these water molecules, being the adventurous types, start to travel across the semipermeable membrane, from Solution B into Solution A. It’s like a slow-motion migration, a gentle tide of hydration moving towards the more concentrated solution. They’re not trying to get rid of the sugar; they’re just trying to dilute it. It’s the ultimate "let’s all chill out together" approach.
This process is called osmosis. Isn't that a fun word? Osmosis! Say it with me. Osmosis! It’s like the universe's way of saying, "Okay, you've got too much of a good thing on one side, so we're going to send some of the good stuff (water!) over there to even it out."
What’s the end result of this watery adventure? Well, over time, the volume of Solution A will increase. It’ll get bigger and bigger, like a balloon slowly being inflated. The concentration of sugar in Solution A will decrease, as it’s getting more and more water. Meanwhile, Solution B will shrink, as it’s losing its precious water molecules. It’s a bit like when you’re at a party and everyone is crowding around the snack table, leaving the quiet corners empty. The water is essentially migrating to where there's more "solute" (the dissolved stuff, like sugar) to try and make things more balanced.
This might sound like a purely theoretical, lab-coat-wearing concept, but think about your body again. Your red blood cells, for example, are tiny little guys floating around in your bloodstream. The liquid inside them is like a specific solution, and the liquid outside them is also a solution. If the balance of water and solutes outside the cell changes too drastically, those little red blood cells can get into trouble.
Imagine you’re swimming in a super-salty ocean. Your body is designed to maintain a very specific internal salt concentration. If you were to drink a lot of that salty ocean water, your body would have to work overtime to get rid of the excess salt. This is because the concentration of salt outside your cells would become higher than inside. Then, water would start to leave your cells, trying to balance out that salty external environment. It's like your cells are saying, "Whoa, too much salt out here! Let's bail!" This can lead to dehydration, which is why you’re supposed to drink fresh water, not ocean water. The semipermeable membrane of your cells is doing its job, but it can only handle so much imbalance!
On the flip side, if you were in a situation where the liquid outside your cells had a much lower salt concentration than inside, water would rush into your cells. This is like putting a plant in plain water when it’s used to a certain soil moisture level. The water goes where the "stuff" is more concentrated, trying to dilute it. In extreme cases, cells can actually swell up and burst, like an overfilled water balloon. Not a good look for your cells, I assure you.
Even something as simple as soaking your feet in a warm bath involves a little bit of this magic. The skin on your feet, while not as permeable as a cell membrane, does allow some water molecules to enter, making your fingers and toes all wrinkly. It’s not necessarily a massive influx, but it's a demonstration of water moving to areas of higher solute concentration (your cells) from an area of lower solute concentration (the bathwater).
So, when we talk about a semipermeable membrane being placed between two solutions, we're essentially setting up a controlled environment to observe this natural balancing act. It's about the flow of water from an area where it's in higher concentration (and therefore the solute is in lower concentration) to an area where it's in lower concentration (and the solute is in higher concentration). It's a constant, quiet hustle happening all around us, from the microscopic world of our cells to the grander scale of ecosystems.
Think of it like a highly exclusive buffet. Solution A has a huge line for the lobster, while Solution B just has a single breadstick. The water molecules, being the sensible party-goers, will naturally migrate towards the lobster buffet to help thin out the crowd and make the ratio of people to lobster a bit more reasonable. They aren't trying to eat the lobster; they're just trying to make the whole situation less overwhelming for everyone involved.
The membrane, in this scenario, is like the velvet rope. It allows the general public (water) to mingle and move about freely, but it keeps the VIP guests (sugar molecules) contained to their designated area. It’s all about regulating the flow and maintaining a certain order, albeit a dynamic one. It’s the ultimate control freak, but in a necessary, life-sustaining way.
So, the next time you see water moving, or your body working its magic to keep everything balanced, remember the semipermeable membrane. It’s the unsung hero, the picky but essential gatekeeper, making sure that life’s intricate processes happen just right. It’s a fundamental principle, and understanding it helps us appreciate the amazing complexity of even the simplest things. And who knew science could be so… wet?
