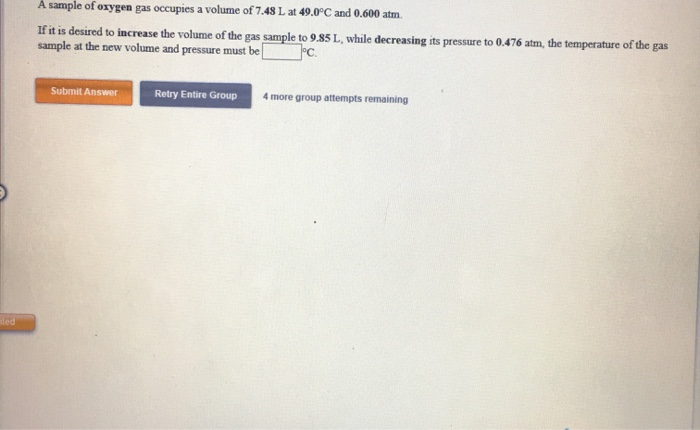
A Sample Of Oxygen Gas Occupies A Volume Of 250

I remember this one time, ages ago, I was trying to impress this girl with my, shall we say, limited knowledge of science. We were picnicking by a lake, and she pointed at a massive, fluffy cloud drifting lazily overhead. "Isn't it amazing," she’d sighed dreamily, "how much stuff is up there?" And me, wanting to sound super smart, I puffed out my chest and declared, "Oh yeah, that's mostly just… air. You know, nitrogen and oxygen. Mostly nitrogen, though. Oxygen's kinda sparse."
She just blinked. The dreaminess evaporated faster than a puddle in the Sahara. Clearly, my attempt at intellectual seduction had backfired spectacularly. The point is, while I might have gotten the basic elements right, my understanding of just how much of anything is actually there was, well, pretty fuzzy. Especially when it comes to gases. They're so… invisible, right? You can't really grasp their bulk, their density, their sheer presence in the same way you can a rock or a loaf of bread. But trust me, even those wispy, ethereal gases take up space. And sometimes, they take up a lot of space. Like, 250 cubic centimeters of it, for example.
So, let's talk about this, shall we? Let's dive into this seemingly innocent little fact: A sample of oxygen gas occupies a volume of 250… wait for it… cubic centimeters. Now, at first glance, that might sound like a tiny amount. I mean, 250 cm³? That’s like… a shot glass, give or take. Or maybe a small, particularly enthusiastic teacup. But here's the kicker: that's oxygen gas. Not liquid. Not solid. But gas.
Think about it. What does "occupies a volume" even mean when you're talking about a gas? Unlike liquids and solids, which have a pretty fixed shape and size, gases are famously unruly. They spread out. They fill whatever container you put them in. They’re like tiny, hyperactive toddlers at a birthday party, bouncing off the walls and generally making their presence known everywhere. So, when we say this sample of oxygen gas occupies 250 cm³, we're essentially talking about the size of the container it's currently residing in. It's the space that those energetic little oxygen molecules have decided to call home, for now.
The Invisible Bulk
It’s easy to underestimate gases. We breathe them in and out without a second thought. We don't see them, we don't feel them in the way we feel a solid object. But they are undeniably there. They have mass. They have volume. And understanding their volume is actually pretty darn important in a whole bunch of scientific and even everyday scenarios. Think about it: when you inflate a balloon, you're not just magically making it bigger; you're adding gas, and that gas is pushing out, expanding to fill the entire balloon. The volume of the balloon is directly related to the amount of gas you’ve shoved into it.
So, this 250 cm³ of oxygen? It's not some magical, self-contained little bubble of oxygen just floating around. It's oxygen that has been confined, or has naturally found itself, within a space that measures 250 cubic centimeters. It's like saying "a bowl of cereal holds 300 grams of cereal." The 300 grams is the cereal, but the "holds" part implies the container. In the case of gases, the container is everything. It's the boundary that defines the volume. Pretty neat, huh? It’s like the gas is saying, "Yeah, I could go anywhere, but I’m chilling in this specific 250 cm³ space right now."

What Determines This Volume?
Now, you might be wondering, what makes that oxygen gas decide to occupy exactly 250 cm³? Is it feeling particularly generous? Is it a greedy gas? Not quite. The volume a gas occupies is super sensitive to its surroundings. It’s like a teenager’s mood – it can change on a dime.
The two big players here are temperature and pressure. Imagine our 250 cm³ of oxygen is in a sealed container. If we heat it up, those oxygen molecules start zipping around faster. They’re bumping into each other and the container walls with more oomph. If the container is rigid, the pressure inside will go up. But if the container can expand, like a balloon, the gas will push outwards and increase its volume to maintain a more or less constant pressure. It’s trying to escape that increased kinetic energy by spreading out.
Conversely, if we cool the gas down, those molecules slow down. They’re less energetic, they don’t push as hard. In an expandable container, the volume would decrease. Think of a deflated beach ball on a cold day – it’s definitely smaller than when it’s warm. It’s all about those tiny molecular collisions and how much space they need to exert a certain pressure.
And then there's pressure. If you were to, I don't know, squeeze that 250 cm³ of oxygen into a smaller container (assuming you could, of course!), it would still be the same amount of oxygen, but it would now occupy a smaller volume. If you reduced the pressure on it (again, in an expandable container), it would likely expand to fill a larger volume. It’s a delicate dance between the gas’s internal energy and the external forces acting upon it.
So, that 250 cm³ isn't some arbitrary number. It's a snapshot in time, a specific condition. It implies a certain pressure and a certain temperature under which that particular amount of oxygen is behaving. If those conditions change, the volume will likely change too, even if the amount of oxygen remains exactly the same. Fascinating, isn't it? It’s like the gas is constantly saying, "Tell me the temperature and pressure, and I'll tell you how much space I'm taking up!"
The Real Quantity: Moles and Molecules
Now, you might be thinking, "Okay, so it takes up 250 cm³. But how much stuff is actually in there?" That’s where things get a little more interesting, and where we start talking about the actual quantity of gas, rather than just the space it occupies. Because 250 cm³ of oxygen at one temperature and pressure is a very different amount of oxygen at another.
In science, we don’t usually measure gases by their volume alone when we want to know the actual amount of substance. We use something called the mole. A mole is just a ridiculously huge number (Avogadro’s number, to be precise – about 6.022 x 10²³) of particles. These particles could be atoms, molecules, whatever. In our case, they're oxygen molecules (O₂).
So, our 250 cm³ of oxygen gas, under specific conditions, will contain a certain number of moles. What are those specific conditions? Well, the most common set we scientists tend to reference is Standard Temperature and Pressure (STP). STP is usually defined as 0 degrees Celsius (273.15 Kelvin) and 1 atmosphere of pressure. At STP, one mole of any ideal gas occupies a volume of approximately 22.4 liters, which is 22,400 cm³.

Now, let's do a little mental math, or maybe even a quick scribbled calculation on a napkin. If 22,400 cm³ is one mole of gas at STP, how many moles are in our 250 cm³ sample? It would be 250 cm³ / 22,400 cm³/mole. That’s… a pretty small number of moles. Around 0.011 moles. That’s roughly 0.011 multiplied by 6.022 x 10²³ molecules. Still a lot of molecules, mind you! We’re talking about billions and billions of tiny O₂ particles, all zipping around in their little 250 cm³ space.
But here’s the catch: if our 250 cm³ of oxygen isn't at STP, then that 0.011 moles isn't accurate. If it’s warmer, or the pressure is higher, the molecules are more squished together, and the same number of moles will occupy a smaller volume. If it’s colder, or the pressure is lower, they’ll spread out and occupy a larger volume. The volume is the consequence of the amount of gas and the conditions it's under.
Why Does This Matter?
You might be asking yourself, "Okay, this is all very theoretical. Why should I care about 250 cm³ of oxygen gas occupying a volume?" Well, believe it or not, this concept pops up in more places than you might think. It’s the foundation for understanding gas laws, which are crucial in fields like chemistry, physics, and even engineering.
Think about engines. They rely on the expansion and contraction of gases when heated and cooled to generate power. Understanding how much volume a certain amount of gas takes up under different conditions is vital for designing efficient engines. Or consider medical applications. When a patient is given oxygen therapy, doctors need to know precisely how much oxygen they're administering. This often involves understanding gas volumes and pressures.

Even in your own home, you’re indirectly interacting with these principles. The gas in your stove, for instance, is delivered at a specific pressure and flows into your burners, where it mixes with air and burns. The efficiency and safety of that process depend on understanding the behavior of gases.
And let’s not forget the environment! Atmospheric science deals with vast quantities of gases. Understanding how temperature and pressure affect the volume these gases occupy helps us model weather patterns, climate change, and air pollution. That seemingly insignificant 250 cm³ is just a tiny microcosm of these much larger, more complex systems.
So, the next time you see a fluffy cloud, or take a deep breath, or even just see a gas gauge, remember that gases aren’t just empty space. They have substance, they have volume, and their behavior is governed by fascinating scientific principles. That 250 cm³ of oxygen? It’s a reminder that even the invisible can have a tangible presence, and understanding its volume is the first step to understanding the amazing world of gases.
It’s a small number, 250 cm³, but it represents a whole lot of tiny, energetic oxygen molecules doing their thing. And that, my friends, is pretty cool. Makes you wonder what other seemingly small numbers in science are actually holding onto a universe of information, doesn’t it? Keep looking, keep questioning. That's how we all learn, one tiny volume of gas at a time. And hey, next time you're trying to impress someone with science, maybe start with something a bit more tangible than clouds. Unless, of course, you can accurately explain the volume occupied by water vapor molecules. Then, by all means, go for it!
