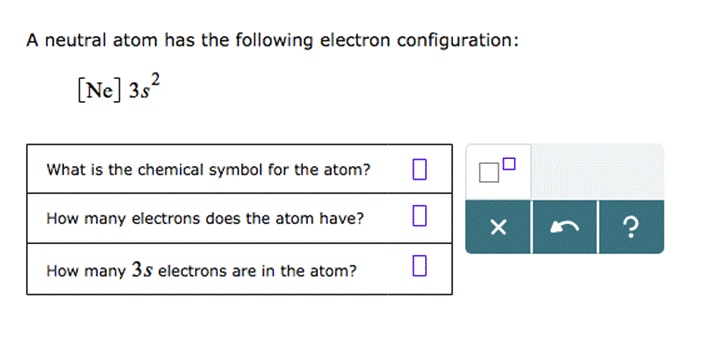
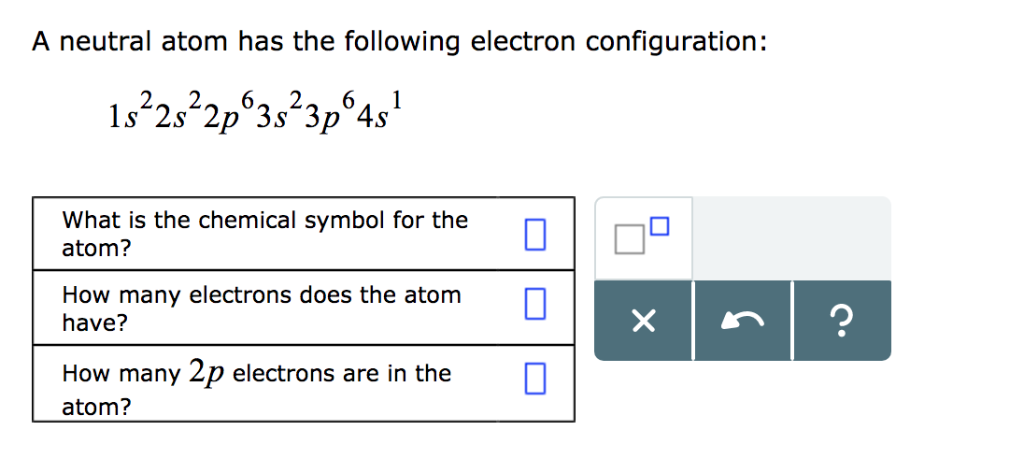
A Neutral Atom Has The Following Electron Configuration:

Hey there, curious minds! Ever feel like life’s just a big, chaotic jumble of stuff? Well, guess what? Even the teeniest, tiniest bits of the universe have their own super-organized systems. And today, we’re diving into one of the coolest: the electron configuration of a neutral atom! Sounds sci-fi, right? But trust me, it’s way more fascinating and, dare I say, fun than it sounds.
So, what’s a neutral atom, you ask? Imagine a super-balanced tiny little being. It’s got a nucleus, which is like its cozy home in the center, packed with positively charged protons. And zipping around this home, like hyperactive toddlers, are negatively charged electrons. When we say an atom is "neutral," it means it’s got the perfect balance. The number of positive protons exactly matches the number of negative electrons. No drama, no tug-of-war, just pure atomic equilibrium. How neat is that?
Now, these electrons, they’re not just randomly bouncing around. Oh no! They’re like little organized citizens, each with their own assigned “address” or energy level. Think of it like a really swanky hotel with different floors, each floor offering a different level of luxury. The closer the electrons are to the nucleus (that central hub), the more they’re “attracted” to it, and the lower their energy. These are the VIPs, the ones who get the prime real estate!
This is where the magic of electron configuration comes in. It’s basically the atomic version of a meticulously crafted seating chart for a very important party. It tells us exactly where each electron hangs out, in which “shell” or energy level, and even in which specific “orbital” within that shell. And these orbitals? They’re not just empty spaces; they have specific shapes, like little clouds or dumbbells, where the electrons are most likely to be found. It’s like assigning each guest their own perfectly shaped, comfortable lounge chair!
The Shell Game: A Symphony of Energy Levels
Let’s break down these “shells.” We start with the shell closest to the nucleus, which is the first energy level. This little guy can only hold a maximum of two electrons. Think of it as a tiny, exclusive studio apartment. Only the most energetic (or least energetic, depending on how you look at it!) can snag a spot there. These electrons are super close to the action, feeling that positive pull from the nucleus quite strongly.
Then we move to the second energy level. This one’s a bit more spacious, a whole floor dedicated to more electrons. It can hold up to eight electrons. It’s like upgrading to a cozy one-bedroom! This level has different types of orbitals, sort of like different types of rooms within the apartment. We’ve got our “s” orbitals, which are nice and spherical, and our “p” orbitals, which are a bit more like two balloons tied together. The electrons fill these up in a specific order, always seeking the lowest energy available.
And it keeps going! The third energy level can hold even more, up to 18 electrons. This is like a sprawling penthouse suite with multiple rooms and even different kinds of views! We see the introduction of “d” orbitals here, which have even more complex shapes. It’s like the atom is building its own elaborate mansion, meticulously designing each room for its electron residents.
You get the idea, right? It’s all about filling up these energy levels, starting from the lowest and working our way up. It’s a fundamental principle in chemistry, this idea of atoms wanting to achieve the most stable, lowest energy state. It’s like us, wanting to relax on the comfiest couch after a long day. Atoms are just doing it on a much tinier, yet infinitely more complex, scale.
Why Should You Care About Electron Arrangements?
Okay, so atoms have their electrons all neatly arranged. Big deal, right? Wrong! This is where it gets really exciting and makes the world around you come alive. The way these electrons are arranged dictates everything about how an atom behaves. It’s like the personality of the atom!
Think about it: if an atom has a full outer shell of electrons, it’s like it’s wearing a perfectly fitting suit. It’s happy and stable. It doesn't really want to interact with other atoms because it’s already got everything it needs. These are the noble gases, like Helium or Neon – you know, the stuff that makes balloons float and signs glow! They’re the introverts of the atomic world, content in their own company.

But what if an atom has an almost full outer shell? Or just one or two electrons hanging out there? This atom is like someone who’s just missing that one ingredient for a perfect recipe. It's eager to interact, to bond with other atoms. It’s looking for a partner to share those electrons, to achieve that coveted full outer shell. This is how chemical bonds are formed, the very glue that holds everything together, from the water you drink to the phone you’re probably reading this on!
The electron configuration determines whether an atom will be a metal, a nonmetal, or something in between. It dictates its reactivity, its color, its conductivity – basically, its entire identity and how it interacts with the universe. It’s the reason why iron rusts, why salt dissolves in water, and why the sun shines! It’s all thanks to these meticulously organized electrons.

Imagine a dance floor. The electrons are the dancers, and the orbitals are the designated spots. Some dancers are content in their solo spots, while others are looking for partners to create a beautiful, synchronized routine. The entire dance, the entire chemical reaction, is orchestrated by the arrangement of these electrons. Pretty cool, huh?
So, next time you look at something – a leaf, a rock, even the air you breathe – remember that it’s all made of atoms, and those atoms have a precise, elegant system of electron arrangement. It’s a testament to the underlying order and beauty in the universe. It’s a reminder that even in the smallest things, there’s a story of balance, energy, and interaction waiting to be understood.
Don't be intimidated by the fancy terms. The beauty of science is that it’s all about uncovering these fascinating patterns. Understanding electron configuration isn't just about memorizing rules; it’s about appreciating the intricate dance of matter that makes up everything we know. So, go forth, be curious, and let the amazing world of atomic structure inspire your sense of wonder. Who knows what other amazing secrets the universe is ready to share with you?
