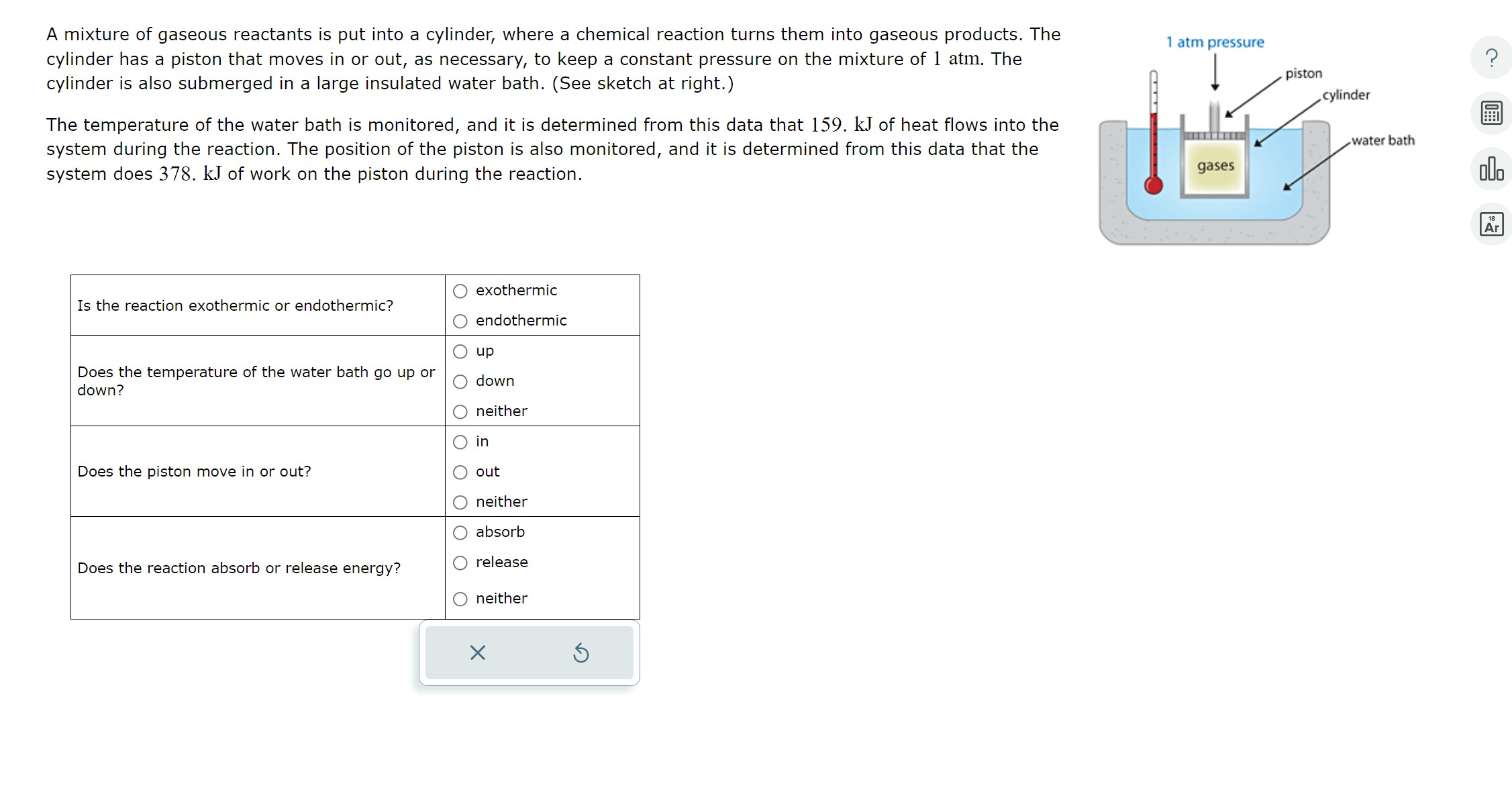
A Mixture Of Gaseous Reactants Is Put Into A Cylinder

Imagine this. You’ve got a bunch of invisible stuff. We’re talking about gases, the floaty, wispy kind. These aren't your everyday breathers, though. These are the kind that like to party.
So, we take these particular gases. And we decide, "Hey, let’s put them all together." It’s like throwing a surprise party for molecules. They're all just chilling, minding their own business.
Then, poof, they’re all crammed into a shiny new cylinder. Think of it as a fancy, metal hotel for gases. They’ve just checked in, and they have no idea what’s about to happen.
This cylinder is usually pretty sturdy. It’s built to handle a lot. Because, let me tell you, these gases are about to get real cozy. Maybe a little too cozy.
They’re all bouncing around. Bumping into each other. It’s a molecular mosh pit in there. They’ve got energy, you see. Lots of it.
Now, the fun part. These aren’t just any random gases. These are reactants. That’s a fancy word for "gases that are up for some drama." They’re looking for a reaction, literally.
When you mix these specific gaseous reactants together in a cylinder, something’s gotta give. It’s a bit like mixing volatile personalities. You know, the ones who just can't help but stir the pot.
They’re all in their little cylinder home. Suddenly, they realize they’re not alone. And some of them have very different ideas about how to behave.
It’s a delicate balance, you know? Like a tiny chemical cocktail. You’ve got your hydrogen, maybe. And your oxygen. They seem innocent enough, right?
But put them in a cylinder, give them a little nudge, and bam. Things can get interesting. Suddenly, they’re not just gases anymore. They’re a team, working on a project.
And this project? It often involves making a lot of noise. Or heat. Or both. It’s the gaseous equivalent of a toddler discovering finger paints and a drum set simultaneously.
You see, these reactants have a destiny. They're destined to change. To transform. To become something new and exciting. Or sometimes, just something very wet.
It's like a magical transformation, but with more fizz. And maybe a bit more smoke. Definitely more scientific jargon, if you’re into that. But at its heart, it’s just stuff changing.
Think about it. You have these invisible ingredients. You seal them up tight. And then, the universe decides to play chef.
The cylinder is the pot. The gases are the ingredients. And the "reaction" is the cooking process.

Sometimes, the cooking is gentle. A slow simmer. A subtle change. You might not even notice it happening at first.
Other times, though? Oh boy. It’s a full-blown culinary explosion. A chemical firework display.
My unpopular opinion? This whole "putting gaseous reactants into a cylinder" thing is way more thrilling than most people give it credit for.
We’re so busy with our spreadsheets and our traffic jams. We forget about the amazing, microscopic drama happening all around us. Or, in this case, inside a cylinder.
It’s like a tiny, contained universe of possibility. Where elements decide to team up and create new compounds. It’s the ultimate collaboration.
Imagine the conversations those molecules are having. "Hey, Bob, you wanna combine forces and make some water?" "Sure, Alice, but it might get a little… energetic."
And the cylinder? It’s the safe space for this energetic collaboration. It holds it all in. Like a very strict bouncer at a wild party.
It prevents the explosive personalities from escaping. It keeps the whole show contained. Which is probably a good thing for all of us living outside the cylinder.
But the magic is happening within those metal walls. A silent, unseen transformation.
You might have methane and oxygen in there. Just sitting. Waiting.
And then, a spark. A little bit of heat. And whoosh. You’ve got water and carbon dioxide. Life-giving stuff, in its own way.
It’s a chemical dance. A molecular ballet. But instead of tutus, they’ve got electrons. And instead of violins, they’ve got… well, they don’t have sounds, but you get the idea.

The beauty of it is the surprise. You can set up the conditions. You can introduce the players. But you never quite know the exact flavor of the reaction until it happens.
Will it be a gentle puff of steam? Or a mini-explosion that rattles the whole lab?
The variables are endless. The possibilities are vast.
It’s a fundamental aspect of chemistry, sure. But let’s not reduce it to a dry textbook chapter.
It’s a captivating display of matter’s potential. A glimpse into the hidden forces that shape our world.
Think of the scientists. They’re like cosmic matchmakers. They’re introducing these gaseous souls, hoping for a beautiful union.
And when it works, it’s beautiful. When it really works, it’s spectacular.
It’s the ultimate experiment in chemistry. A contained chaos. A controlled wildness.
So next time you hear about gaseous reactants being put into a cylinder, don’t just think of boring science.
Think of the potential. Think of the surprise. Think of the tiny, invisible world getting ready to put on a show.
It’s a miniature universe, brewing and bubbling. And all we have to do is appreciate the unseen spectacle.
It’s the kind of thing that makes you wonder about all the other invisible processes happening around us.

The silent transformations that are constantly at play.
And it all starts with a simple act: putting some reactive gases into a cylinder.
It’s the beginning of a beautiful, and sometimes explosive, friendship.
And honestly? I think that’s pretty darn cool.
It’s a reminder that even in the most mundane-looking containers, extraordinary things can occur.
It’s the thrill of the unknown, sealed in metal.
The quiet hum of potential energy, just waiting for its cue.
So, let’s celebrate these cylinders. These vessels of transformation.
These unassuming stages for the grandest of chemical performances.
It’s chemistry, but make it exciting. Because, let’s be honest, it is.
These gaseous reactants, mingling and merging. It’s a story unfolding.
And the cylinder is just the cover of the book.
It’s where the adventure truly begins.
And who knows what wonders they’ll create next?
The possibilities are, quite literally, explosive.
So, here’s to the cylinders. And the incredible, invisible dancers within.
They’re the unsung heroes of chemical change. And they deserve a little applause.
Even if the applause is just a silent nod of appreciation.
For the magic that happens when gases get together.
In their shiny, metal home.
Just waiting for their moment.
It’s a beautiful thing to witness.
Even when you can’t see it.
And that’s the true marvel.
Isn't it?
