A Hydrogen Filled Balloon Was Ignited And 1.50g
So, picture this: you've got a balloon, right? Not just any balloon, mind you. This one's stuffed to the gills with hydrogen. Now, some people might think, "Ooh, science experiment!" and others might think, "Uh oh, this can't be good." And to those latter folks, I say, you are absolutely correct.
We're talking about a tiny bit of gas, just a minuscule 1.50 grams of the stuff. That sounds like, what, the weight of a couple of paperclips? Less, even. You could lose that much pocket lint in a week. But this seemingly insignificant amount of hydrogen, when given a little oomph of ignition, decided to put on a show. A rather energetic show, as it turns out.
Now, you might be wondering, "What's the big deal about 1.50 grams of hydrogen?" Well, my friends, hydrogen is like the party animal of the gas world. It's incredibly lightweight, making it perfect for those big, majestic blimps (though maybe not ideal for the Titanic of blimps, hindsight being 20/20). But it's also, shall we say, highly combustible. Think of it as a tiny, enthusiastic pyromaniac in a balloon suit.
When you introduce a spark – a little lick of flame, a stray static shock, or perhaps just a poorly timed sneeze near an open flame – to hydrogen, it doesn't just go "poof." Oh no. It goes WHOOSH! BAM! BOOM! It's a chemical reaction that's been compared to a miniature supernova, which is a bit dramatic, but honestly, when you see it, you might just agree. It's like the universe decided to create a tiny, personal fireworks display, just for your amusement (or terror).
The science behind it is actually pretty neat, if you ignore the slight urge to duck for cover. Hydrogen (H₂) is made of two hydrogen atoms. When it meets oxygen (O₂) from the air, and gets a little nudge from some heat (the ignition), they get very excited. They decide to team up and form water (H₂O). But the process of them becoming water is like a tiny explosion of energy. It's a whole lot of heat and light being released in a blink-and-you'll-miss-it moment. Like a scientific speed dating event gone spectacularly wrong… or right, depending on your perspective.

So, our 1.50 grams of hydrogen, thinking it's going to have a nice, calm float around the room, suddenly gets introduced to a spark. And what happens? It's like the gas decided to spontaneously combust, but in the most dramatic way possible. Imagine a tiny, contained explosion happening right there. Not enough to level a city block, thankfully, but definitely enough to make your eyebrows do a little dance.
It’s a reminder that even the smallest things can have a big impact. That 1.50 grams of hydrogen is a testament to the power locked within seemingly simple substances. It's the difference between a gentle breeze and a hurricane, all wrapped up in a latex sphere. It's the reason we don't let kids play with hydrogen balloons near open fires, unless we're aiming for a surprisingly quick (and possibly singed) science lesson.

Think about it: if you took that 1.50 grams of hydrogen and put it into a fuel cell, it could power a small device for a decent amount of time. It's clean energy! But if you light it? It’s a fleeting, fiery spectacle. It’s like the difference between a well-behaved citizen and a rock star trashing a hotel room. Same material, wildly different outcomes.
And the speed of it! This isn't a slow burn, folks. This is an instantaneous combustion. It’s faster than you can say “safety goggles.” The flame front travels through the hydrogen at an incredible speed, consuming the gas and releasing all that stored energy. It’s a race against time, and in this case, the flame wins. Every single time. It’s the ultimate "carpe diem" moment for a gas molecule.
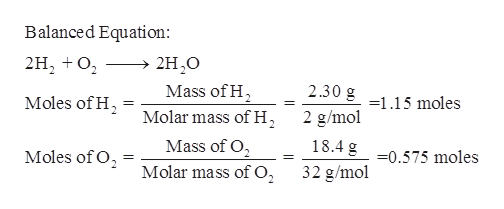
It’s also worth noting that the sound! Hydrogen explosions, even small ones, can be surprisingly loud. It’s not a gentle pop; it’s more of a sharp crack or a surprising bang. It’s the kind of sound that makes you jump, spill your coffee, and then immediately want to tell everyone about the amazing thing you just witnessed. It’s the sound of pure, unadulterated chemical excitement.
So, next time you see a hydrogen balloon, just remember the potential. That seemingly innocent, floating bubble is a tiny powerhouse of explosive energy, just waiting for its moment. And while 1.50 grams might sound trivial, in the world of chemistry, it’s enough to put on a rather memorable, and quite frankly, entertaining, show. Just make sure you’re watching from a safe distance, and maybe have a glass of water handy. For the balloon, you know, to help it cool down… eventually.
