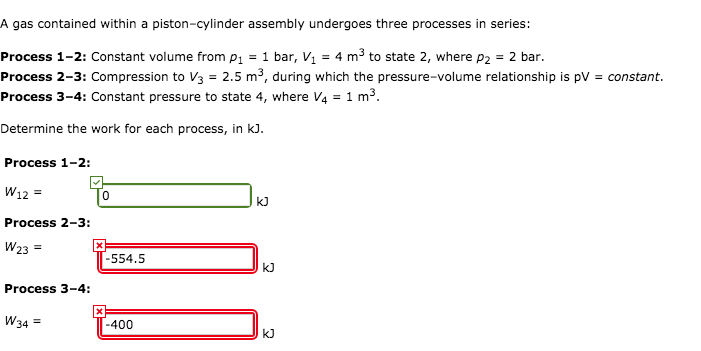
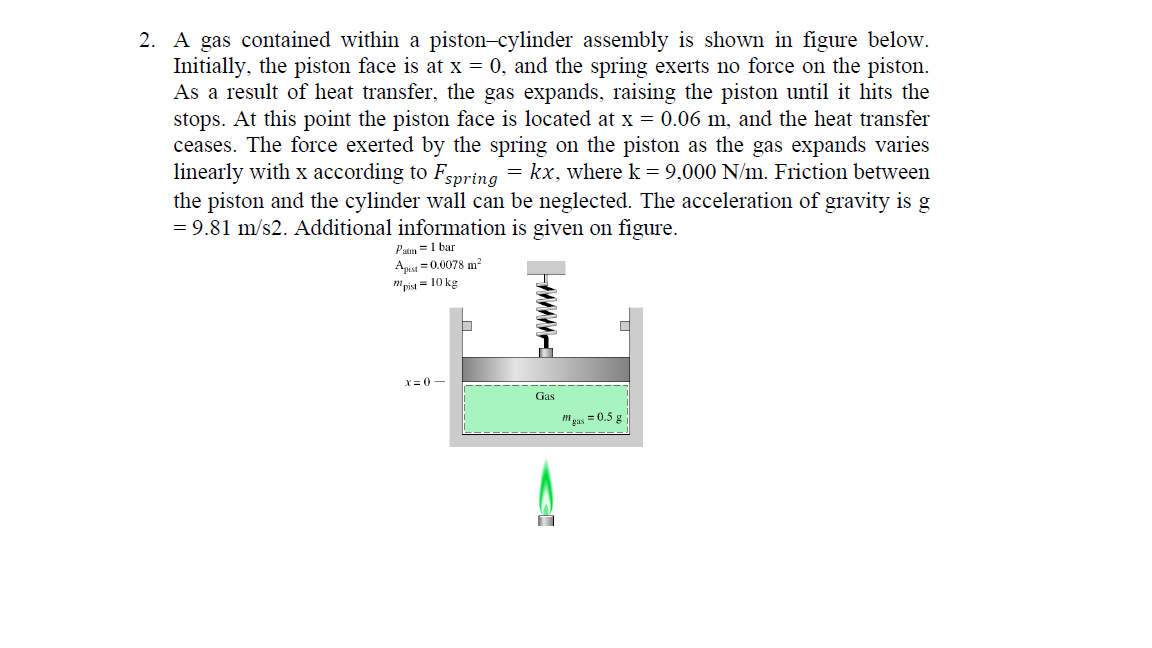
A Gas Contained Within A Piston Cylinder Assembly

Hey there, science curious friends! Ever feel like life’s a bit… well, contained? Like you're stuck in a rut, going through the motions? What if I told you a little secret about a tiny, everyday hero that’s all about expanding possibilities? Yep, we're talking about a gas, chilling out in a piston-cylinder assembly. Sounds a bit nerdy, right? But stick with me, because this seemingly simple setup is actually a miniature powerhouse of wonder, and understanding it might just inject a little extra oomph into your day!
Think about it. What’s a gas? It's like that energetic friend who just can't sit still, bouncing around, filling up every nook and cranny. Now, imagine that energetic friend crammed into a very polite, very orderly container – our piston-cylinder assembly. It's like a snug little apartment for our gas molecules, but with a crucial difference: this apartment has a movable wall!
This movable wall is our piston. It’s the gatekeeper, the control freak, the ultimate influencer of our gas's mood. Our gas, in its infinite, bouncy enthusiasm, is constantly bumping and pushing against the walls of its cylinder, and yes, that includes our piston. It’s like a never-ending game of molecular tag!
So, what happens when this gas gets a little too excited? Maybe we give it some heat, like a tiny molecular spa day. What do those bouncy molecules do? They get even more bouncy! They start zipping around faster, colliding with more force. And what happens when they collide with that piston? They push it!
Suddenly, our contained gas isn't so contained anymore. It's doing something amazing: it's doing work. That’s right! That little push on the piston? That’s the gas literally moving something. It's like a miniature, invisible butler, tidying up its space by nudging the furniture (the piston!).
Now, you might be thinking, "Okay, a gas pushing a piston. So what?" But this is where the magic starts to unfurl, my friends. This simple interaction is the heart and soul of so many things we take for granted. Ever ridden in a car? Driven by that iconic piston-cylinder setup, humming away under the hood! That gas, expanding with the controlled explosions of fuel, is what propels you down the road.
Think about the humble bicycle pump. It’s a piston-cylinder assembly in action! You're not just pushing air; you're compressing that gas. You're squishing those bouncy molecules closer and closer together. And what happens when you do that? They get a little mad, a little frustrated. They push back even harder! This increase in pressure is what gives your tires that satisfying firmness, ready to roll.
It’s all about the relationship between pressure, volume, and temperature. These are the three musketeers of the gas world in our piston-cylinder scenario. If you change one, the others often have a little dance to follow. It’s a delicate, dynamic equilibrium, like a perfectly choreographed ballet.

Let’s say you have a fixed amount of gas, and you suddenly heat it up inside the cylinder. If the piston is free to move, what do you think happens? Yep, you guessed it! The gas gets all excited, expands, and pushes that piston outwards. The volume increases. It’s the gas saying, "Woah, too much energy in here! Gotta spread out!"
Conversely, if you push the piston in, you're reducing the volume. You're making those gas molecules hug each other a bit tighter. This inevitably leads to an increase in pressure. It's the gas whispering, "Hey, give us some space!"
This dance of pressure and volume is fundamental to how engines work. In a gasoline engine, for example, a tiny spark ignites a mixture of fuel and air. This causes a rapid expansion of hot gases, pushing the piston down with incredible force. This linear motion is then converted into rotational motion, ultimately turning your wheels.

It's like a tiny, controlled explosion having a massive impact. And it's all thanks to the fundamental properties of gases and the clever design of a piston-cylinder assembly. It’s a testament to how simple principles can lead to incredibly complex and powerful outcomes.
But it’s not just about cars and pumps. Think about steam engines! Old school, right? But the principle is the same. Water is heated, turns into steam (a gas!), expands, and pushes a piston. This powered everything from trains to factories in a bygone era, shaping the world as we know it.
Even in your own body, there are analogies. Your lungs are like little bellows, acting like piston-cylinder assemblies to bring air in and out. The diaphragm muscle contracts and relaxes, changing the volume of your chest cavity, and the air, being a gas, does the rest!
So, the next time you hear the hum of an engine, or feel the puff of air from a pump, take a moment to appreciate the unsung hero: the gas in its piston-cylinder home. It’s a constant reminder that even the most seemingly ordinary things are bursting with potential. They’re silently, tirelessly working to make our lives easier, faster, and more fun.
This isn't just abstract physics; it's the engine of innovation, the spark of progress. It’s about harnessing the power of simple elements to achieve extraordinary feats. It’s about understanding how things work, and in doing so, unlocking a deeper appreciation for the world around us.
Isn’t that inspiring? The fact that a bunch of invisible molecules, confined in a metal tube, can be the driving force behind so much of our modern world? It’s a beautiful illustration of how understanding the fundamentals can lead to incredible inventions and advancements. So, don't be shy. Dive a little deeper! Explore the world of thermodynamics, of mechanics, of how these seemingly simple systems are the building blocks of our technological marvels. You might just find a whole new universe of fun and fascination waiting for you!
