A Galvanic Cell Is Powered By The Following Redox Reaction
Ever wondered how those little batteries in your remote or even your phone actually work? It’s not magic, although sometimes it feels like it, right? It’s all thanks to something called a galvanic cell, and the secret sauce behind it is a fascinating dance called a redox reaction. Think of it as a chemical tug-of-war, where electrons are the prize!
So, what exactly powers a galvanic cell? It's this very same redox reaction. Pretty neat, huh? But what’s a redox reaction, and why is it so cool that it can make things light up and buzz?
The "Redox" Reveal: It's All About Electrons
Let's break down this fancy word, "redox." It’s actually a mashup of two words: reduction and oxidation. Don't let the big sciencey names scare you; it's actually quite straightforward once you get the gist.
Imagine a couple of friends, let’s call them Allie and Ed. Allie is feeling generous and has an extra shiny toy (an electron) she’s willing to give away. Ed, on the other hand, is eager to get his hands on a new toy. So, Allie loses her toy, and Ed gains it. In the world of chemistry:
- When something loses electrons, we say it gets oxidized. Think of Allie saying, "Oh, I oxidized my electron!"
- When something gains electrons, we say it gets reduced. Ed exclaims, "Hooray, I reduced the electron count of that other guy by gaining one!"
The key thing to remember is that these two processes, oxidation and reduction, always happen together. You can't have one without the other, just like you can't have a tug-of-war with only one person pulling. It's a complete exchange, a chemical hand-off.

From Tug-of-War to Tiny Powerhouse
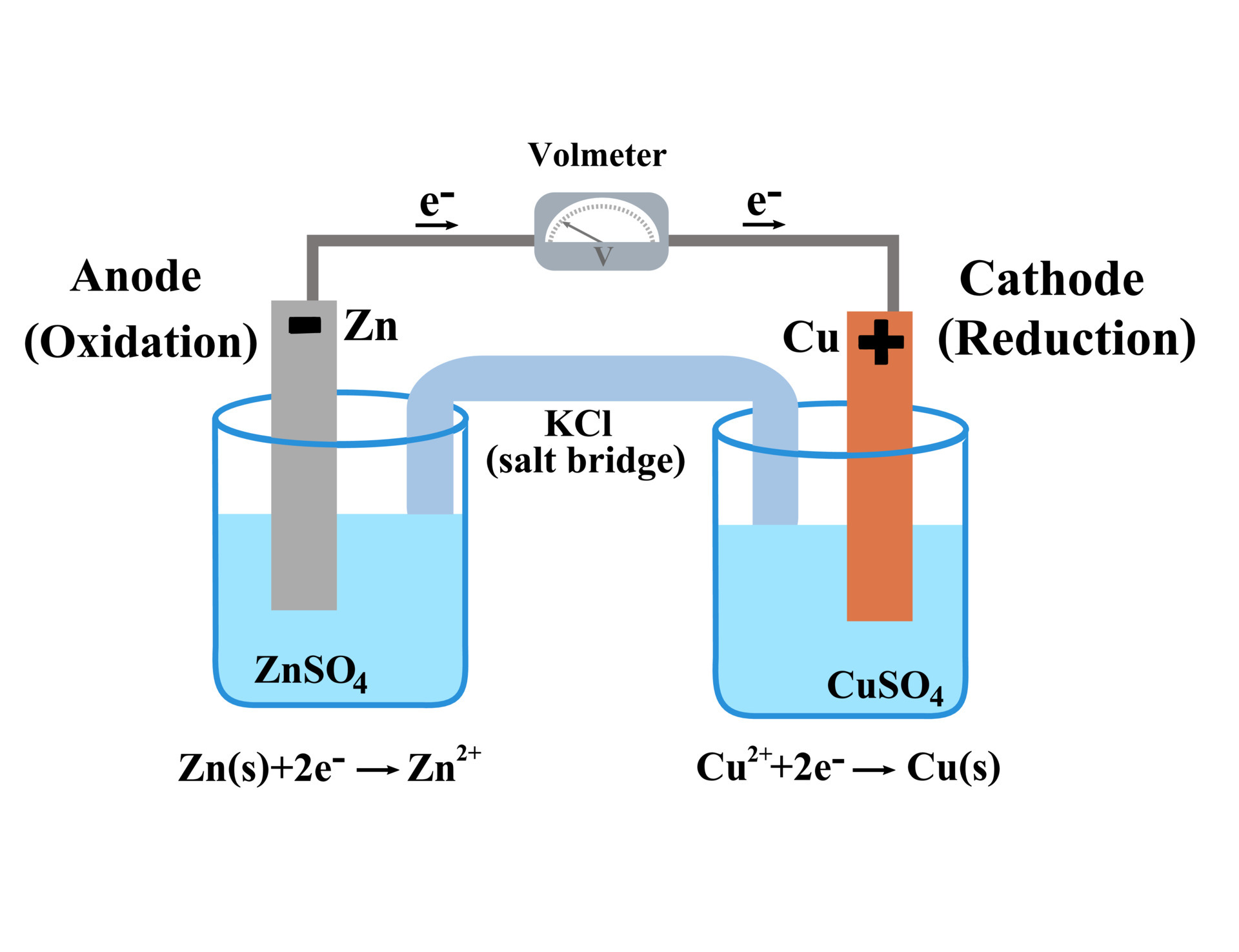
Now, how does this electron-swapping action turn into power? Well, a galvanic cell is designed to make this redox reaction happen in a very specific way. It separates the "givers" and "receivers" of electrons into different locations, called half-cells.
Think of it like this: you have two separate ponds. In one pond, there are lots of floating toys (electrons) that are easily tossed around. In the other pond, there are kids really, really wanting those toys. If you just let them swim between the ponds, chaos! But what if you build a special bridge that only lets the toys cross, and you can control the flow?
That’s essentially what a galvanic cell does. It uses two different materials, often metals, each eager to either give up or accept electrons. These materials are placed in solutions that allow for the electron transfer.

The crucial part is that there’s a pathway, a salt bridge or a porous barrier, that allows ions to move between the two half-cells. This is super important because it keeps the electrical charge balanced. Without it, the reaction would grind to a halt faster than a toddler after a sugar rush.
The Flow of Electrons is Electricity!
So, you have your electron donors and your electron acceptors, separated but connected by that ion highway. When the circuit is completed (like when you connect your battery to your device), the electrons, which are basically tiny packets of energy, start to flow from the material that’s giving them up (the anode, where oxidation happens) to the material that’s taking them in (the cathode, where reduction happens).

And guess what? This flow of electrons is precisely what we call electricity! It’s that invisible force that powers your gadgets, lights up your room, and keeps your music playing. Pretty mind-blowing when you think about it, isn't it?
It's Like a Chemical Cascade
Imagine a series of dominoes. When you push the first one, it knocks over the next, and then the next, creating a chain reaction. A redox reaction in a galvanic cell is similar. The eagerness of one substance to lose electrons triggers a chain of events that ultimately leads to a steady stream of electrons moving through an external circuit.
Different combinations of materials will have different "pulls" for electrons. Some are like a gentle breeze, while others are a strong gust of wind. The bigger the difference in their electron-attracting power (their electrochemical potential), the more voltage, or "push," the galvanic cell can generate. It’s like having a stronger battery that lasts longer!

Why Is This So Cool?
Beyond just powering our everyday lives, the concept of galvanic cells and redox reactions is foundational to so many things. It's the basis of batteries, of course, but it also plays a role in:
- Corrosion: Ever seen rust on a car? That’s a type of redox reaction in action, just not a controlled one!
- Biological Processes: Your own body uses redox reactions for energy production in a process called cellular respiration. So, you're a walking, talking galvanic cell, in a way!
- Electroplating: This is how we get those shiny chrome finishes on faucets or jewelry. It involves using electricity generated by redox reactions to deposit a thin layer of metal onto another object.
The beauty of a galvanic cell is that it harnesses a natural chemical process and turns it into something useful and controllable. It’s about understanding the fundamental interactions of atoms and molecules and using that knowledge to our advantage.
The Takeaway: A Simple Idea, Big Impact
So, the next time you pop a battery into something, give a little nod to the humble redox reaction. It’s the unsung hero, the silent worker, that’s making it all happen. It's a chemical reaction, yes, but it's a reaction with purpose, a reaction that gives us power. It’s a beautiful example of how the seemingly tiny world of atoms can have a monumental impact on our big, everyday world. Pretty cool, right?
