A Four - Electron Reduction Of Oxygen Occurs Within:

Ever wonder what powers your car, lights up your home, or even keeps you breathing? It’s a whole lot of chemistry happening behind the scenes, and one of the most exciting performances involves oxygen! Today, we’re diving into a fascinating process called the four-electron reduction of oxygen. Don't let the science-y name scare you; it's actually a cornerstone of how we get energy and how our bodies function. Think of it as a tiny, but mighty, energy factory working overtime!
This process is super popular and incredibly useful because it’s the backbone of so many technologies and biological systems that we rely on every single day. From the humble battery in your remote control to the incredibly complex machinery inside your own body, the four-electron reduction of oxygen is a common thread. It's a testament to nature's ingenuity – finding efficient ways to transform energy. It's also a key player in some of the cleanest energy solutions we're developing, making it a hot topic in the world of sustainable technology.
Where the Magic Happens: Batteries and Biology!
So, where exactly does this electrifying reaction take place? Two of the most common and crucial places are within electrochemical cells, like the ones found in batteries, and within the tiny powerhouses of our cells, the mitochondria. These aren't just random locations; they are precisely designed environments where this specific chemical dance can occur safely and effectively, releasing valuable energy.
In the world of batteries, this process is the unsung hero of portable power. Think about your smartphone, your laptop, or even the electric car you might dream of owning. They all rely on a carefully orchestrated chemical reaction where oxygen plays a starring role. In many types of batteries, particularly those used in electric vehicles and rechargeable devices, oxygen from the air is taken in and undergoes a transformation. This transformation is where the "four-electron reduction" comes into play. Essentially, oxygen molecules grab onto four electrons, which are tiny negatively charged particles that carry electrical energy. This grabbing action is what drives the flow of electricity, powering your devices.
The beauty of this reaction is its efficiency. It's a clean way to extract a significant amount of energy from oxygen, a readily available resource.
The benefits of this process in batteries are enormous. It's what allows us to have long-lasting power for our gadgets, to drive cars without burning fossil fuels, and to store renewable energy from solar and wind sources. Without this fundamental chemical step, our modern, connected world would look very different. It’s a key reason why batteries are becoming more powerful, more efficient, and more sustainable.
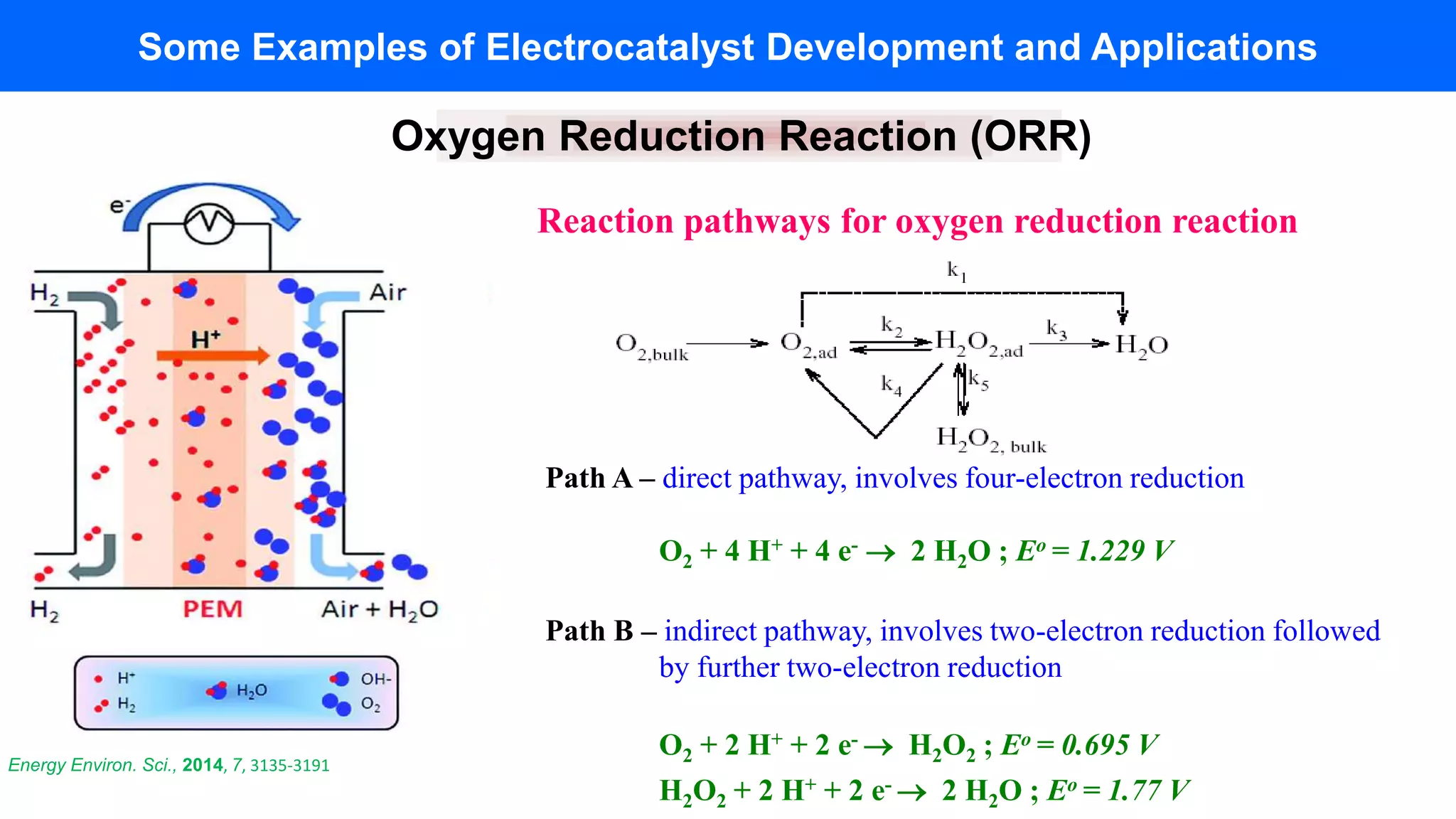
Now, let's shift gears and look inside ourselves. Believe it or not, the very same four-electron reduction of oxygen is happening right now in every single cell of your body, specifically within those amazing organelles called mitochondria. You might have heard of them as the "powerhouses of the cell," and this is why! When you breathe in, you’re taking in oxygen. Your body then uses this oxygen in a complex series of steps, and a crucial part of that process is the reduction of oxygen. In this biological context, the oxygen is used to accept electrons that have been harvested from the food we eat. This electron transfer is the final step in generating adenosine triphosphate (ATP), the energy currency that fuels everything from your muscles to your brain.
The purpose of this in our bodies is, quite simply, to survive and thrive. The energy generated by the four-electron reduction of oxygen allows us to move, think, grow, and repair ourselves. It’s a continuous, vital process happening moment by moment. Without it, our cells wouldn't have the energy to perform their essential functions, and life as we know it wouldn't be possible. The efficiency and elegance of this biological machinery are truly astounding, allowing us to convert chemical energy from food into usable power for our bodies with remarkable precision.

The benefits of understanding and harnessing this process extend beyond just powering our devices and keeping us alive. Scientists are constantly working to improve the efficiency of these reactions, both in batteries and in other applications. For instance, research into more advanced battery materials aims to make them charge faster, hold more power, and last longer, all by optimizing how oxygen participates in the electron transfer. In the realm of biology, understanding mitochondrial function can lead to breakthroughs in treating diseases related to energy metabolism, such as diabetes and neurodegenerative disorders.
So, the next time you charge your phone, take a deep breath, or simply marvel at the energy that powers your own life, remember the incredible work being done by the four-electron reduction of oxygen. It's a fundamental process, happening in both our technology and our very being, that unlocks energy and makes our modern world – and our very existence – possible. It's a tiny chemical reaction with monumental implications!
