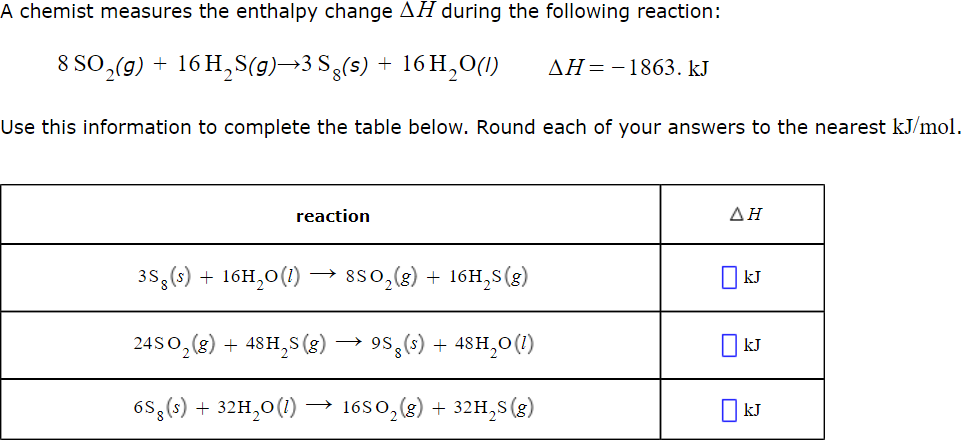
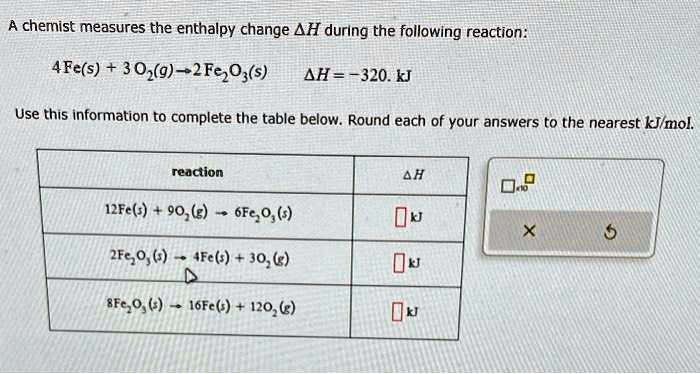
A Chemist Measures The Enthalpy Change During The Following Reaction:

Picture this: a cozy kitchen, the scent of freshly baked cookies wafting through the air. Sounds lovely, right? Now, imagine that same kitchen, but instead of flour and sugar, we're talking about a lab coat and some seriously cool equipment. That’s where our story begins, with a chemist getting down to business, measuring the enthalpy change of a reaction. Think of it as the reaction's “energy footprint” – how much heat it either gives off or sucks up.
It’s not as intimidating as it sounds. In fact, the whole idea of energy exchange is something we deal with every single day. Ever felt that delightful warmth from your morning coffee? That's a tiny, everyday example of enthalpy change in action. Or maybe you've chilled a drink in the freezer, and felt the air around it get a little cooler? Yep, more enthalpy at play.
So, our chemist is essentially doing the same thing, just with a bit more precision and a lot more glassware. They’re observing a chemical reaction and quantifying the energy involved. It’s like being a detective, but instead of clues, they’re looking for changes in temperature.
The Star of the Show: Enthalpy
Let's dive a little deeper into this term, enthalpy. In simple terms, it's the total heat content of a system. When a chemical reaction happens, bonds are broken and new bonds are formed. This process either requires energy (endothermic) or releases energy (exothermic). The enthalpy change, often represented by the Greek letter delta H (ΔH), tells us the net amount of energy that’s swapped between the reaction and its surroundings.
If ΔH is negative, it means the reaction releases heat – we call this an exothermic reaction. Think of a campfire on a chilly evening; it’s giving off heat, making things warmer. That’s exothermic. If ΔH is positive, the reaction absorbs heat from its surroundings – this is an endothermic reaction. Imagine an instant cold pack for an injury; it feels cold because it’s pulling heat into the pack from your skin.
It’s like a scientific mood ring for reactions, telling us whether it’s feeling warm and fuzzy (exothermic) or a bit chilly (endothermic).
The Art and Science of Measurement
How does our chemist actually measure this? They’re likely using a device called a calorimeter. Now, don’t let the fancy name fool you. The basic principle is pretty straightforward.
Imagine a well-insulated container. Inside this container, we have our reactants. The calorimeter is designed to minimize any heat escaping or entering from the outside world, so we can accurately track the heat produced or consumed by the reaction itself.
A common setup involves a beaker or a sealed vessel containing the reactants, submerged in a known amount of water. A thermometer is carefully placed to monitor the water’s temperature. When the reaction kicks off, any heat released will warm the water, and any heat absorbed will cool it down. By measuring the change in the water’s temperature, and knowing how much water there is (and its specific heat capacity – a fun fact for another day!), the chemist can calculate the enthalpy change of the reaction.
It’s a bit like baking a cake. You need the right ingredients (reactants), the right oven temperature (the reaction conditions), and you need to measure how long it bakes (the reaction time) to get the perfect result. In our case, the "perfect result" is a precise measurement of energy.
Why Bother? The Practical Side of Things
You might be thinking, "Okay, but why is measuring enthalpy change so important? Does it affect my daily commute or my Netflix binge?" The answer is a resounding yes, in more ways than you might realize!
Think about energy production. When we burn fuels like natural gas or coal, we're relying on the exothermic enthalpy change to generate heat and electricity. Understanding the precise enthalpy of combustion for these fuels is crucial for designing efficient power plants and managing our energy resources.
In the food industry, enthalpy changes are fundamental to understanding the energy content of food. That calorie count on your favorite snack? It’s directly related to the energy released when your body metabolizes the food, a process involving countless chemical reactions with their own enthalpy changes.

Even in your own kitchen, when you cook, you’re manipulating enthalpy changes. Boiling water for pasta? That’s an endothermic process, requiring heat input. Frying an egg? That’s a series of exothermic reactions that cook the egg. Every time you use heat or cold in cooking, you're interacting with the principles of enthalpy.
And let’s not forget medicine. Many pharmaceutical processes involve precise control of reaction temperatures to ensure the desired product is formed safely and efficiently. Understanding enthalpy changes is key to developing new drugs and manufacturing them on a large scale.
A Glimpse at the Chemistry Behind the Scenes
Let's say our chemist is studying the reaction between an acid and a base, a classic example of an acid-base neutralization. When a strong acid like hydrochloric acid (HCl) reacts with a strong base like sodium hydroxide (NaOH), they form salt and water:
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
This particular reaction is famously exothermic. When these two are mixed, the solution warms up. The calorimeter allows us to measure exactly how much it warms up, giving us a quantitative value for the heat released.

Consider another common reaction: the dissolution of ammonium nitrate in water. This is the key reaction in those instant cold packs. When ammonium nitrate dissolves, it absorbs heat from the surroundings, making the pack feel cold. This is an endothermic process, and its enthalpy change is positive.
So, our chemist is essentially quantifying these everyday phenomena. They’re putting a number on how much “warmth” or “coldness” a reaction brings to the table.
Fun Facts and Cultural Nods
Did you know that the concept of heat itself was once thought to be a fluid called "caloric"? It wasn't until the work of scientists like James Prescott Joule in the 19th century that we understood heat as a form of energy. His experiments on the mechanical equivalent of heat laid the groundwork for modern thermodynamics, including our understanding of enthalpy.
In some cultures, the temperature of food and drink can have symbolic significance. Think of a steaming bowl of ramen on a cold day – it’s not just about physical warmth, but also about comfort and nourishment. Conversely, a chilled glass of wine on a hot summer afternoon is about refreshment and relaxation. These sensory experiences are all tied to the energy transformations happening at a molecular level.
The word "calorie" itself, which we associate with food, comes from the Latin word "calor," meaning heat. It's a direct link back to the fundamental idea of measuring heat energy!
And for all you sci-fi fans out there, think about fictional energy sources or propulsion systems. The underlying principles of how much energy can be extracted or stored often implicitly involve concepts like enthalpy change, even if they're presented with a futuristic twist.
Tips for the Aspiring Home Chemist (with a wink!)
While we’re not encouraging you to start mixing chemicals in your kitchen sink (please, don't!), understanding these principles can be fun. Next time you're cooking, pay attention to the energy changes.
- Observe your cooking: Notice how some things heat up quickly and others take time. What's happening with the energy?
- Think about ice: Why does ice melt? It absorbs heat from the environment, a classic endothermic process.
- Consider your bath: When you add hot water to your bath, you're directly adding energy to the water.
It’s about developing a more mindful awareness of the energy exchanges happening all around us, even in the seemingly mundane tasks of life.
The Heart of the Matter: It’s All About Balance
Our chemist meticulously measuring the enthalpy change of a reaction is, in essence, studying the balance of energy in a chemical transformation. It's a fundamental concept that underpins so much of our modern world, from the power that lights our homes to the food that fuels our bodies.
And that's the beauty of it, isn't it? The complex world of chemistry, with its precise measurements and intricate reactions, is deeply connected to our everyday experiences. Whether it's the warmth of a cup of tea or the chill of a cold pack, we're all, in our own way, experiencing the constant ebb and flow of energy. So, the next time you feel a change in temperature, give a little nod to the chemists out there, diligently measuring the energetic pulse of the universe, one reaction at a time. It’s a reminder that even in the smallest of interactions, there’s a whole lot of energy at play.
