A Cell That Completed The Cell Cycle Without Undergoing Cytokinesis

So, picture this: my friend Sarah, bless her chaotic heart, decided to bake a cake. Not just any cake, mind you, but one of those multi-layered extravaganzas that requires a small army and a degree in structural engineering. She meticulously followed the recipe, mixed, baked, cooled, and then… well, she got distracted. A rogue squirrel had apparently declared war on her bird feeder, and Sarah, ever the protector of innocent avian life, rushed outside with a broom. By the time she returned, the cake batter was still in its bowl, the oven was off, and the layers were… well, they were all there, but somehow still fused together. She’d completed the “baking phase” (let’s call it that for dramatic effect), but the crucial “assembly phase” never happened. It was… a very flat, very monolithic cake.
It sounds ridiculous, I know. A cake stuck in its own batter, so to speak. But it got me thinking. Life, and especially cell life, is all about processes. And sometimes, those processes go a little… wonky. Today, we’re going to dive into a particularly fascinating, and dare I say, slightly monstrous cellular phenomenon: a cell that finished its grand tour of the cell cycle, did all the DNA replication, the chromosome wrangling, the whole nine yards, but then… poof… forgot the grand finale. It skipped the cellular equivalent of cutting the cake. We’re talking about a cell that completed the cell cycle without undergoing cytokinesis.
The Cell Cycle: A Bacterial Dance-Off (But Way More Important)
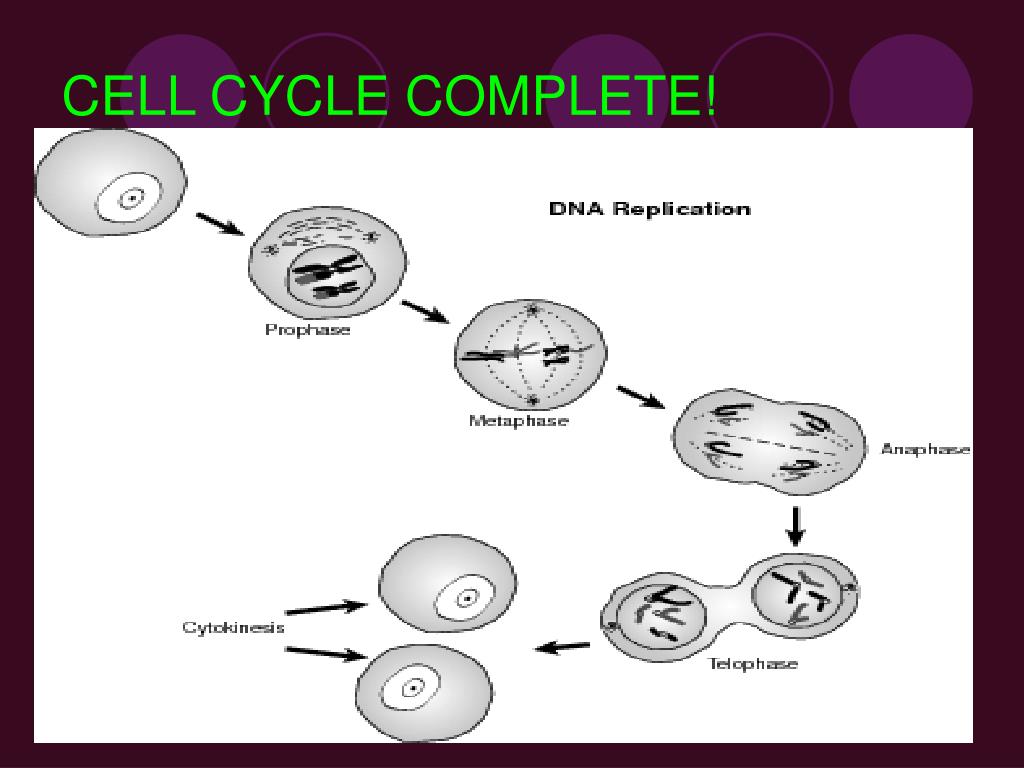
Alright, let's set the stage. You’ve probably heard of the cell cycle, right? It’s basically the life story of a cell, from when it’s born until it divides to make more cells. Think of it like a meticulously choreographed dance. There’s the “G1 phase,” where the cell just lives its life, grows, and gets ready for big things. Then comes “S phase,” the superstar, where it doubles its DNA. This is like copying all your important blueprints before building a second identical house. Crucial stuff!
After S phase, we have “G2 phase,” more growth and final prep. And then, the main event: “M phase,” which stands for mitosis (for the nucleus) and – here’s the kicker – cytokinesis. Mitosis is the part where the duplicated chromosomes get neatly sorted and pulled apart into two future cell nuclei. It’s incredibly precise, like a microscopic ballet where every dancer knows their exact move. Think of it as dividing the blueprints equally between the two new houses.
And then, the grand finale, cytokinesis. This is where the actual physical division happens. The cytoplasm, the cell membrane, all the juicy bits – they get pinched off, or divided, to create two separate, independent daughter cells. It’s the equivalent of actually building those two houses, each with its own foundation, walls, and roof. Two distinct entities, ready to start their own lives.
The Plot Twist: When Cytokinesis Goes AWOL
So, what happens when a cell nails mitosis – all the chromosomes are perfectly segregated, the cell is ready to split – but then forgets or fails to do the actual splitting part? That, my friends, is where things get interesting. Instead of two neat daughter cells, you end up with one massive cell containing two (or more, if this happens repeatedly) nuclei. It’s like Sarah’s cake, all the ingredients baked, but still one giant, undifferentiated mass.
This isn’t just some rare, bizarre anomaly that scientists tut-tut over in their labs (although they do that too, don’t worry). This actually happens in nature, and it has some pretty significant implications. Think about it: a cell that’s supposed to be a certain size, performing specific functions, suddenly becomes a giant, multi-nucleated behemoth. It’s like a single-celled organism deciding to go on a steroid binge. Not exactly ideal for efficient functioning, right?
Why So Big? The Consequences of Skipping the Split
When a cell skips cytokinesis, it’s usually referred to as a multinucleated cell. And the reasons for this can be varied. Sometimes, it's a deliberate developmental strategy. For example, in some tissues, multinucleated cells are supposed to form. Think of your skeletal muscle cells – they’re actually huge, long cells with many nuclei, formed by the fusion of many smaller cells. This allows for coordinated muscle contraction. So, in this case, it’s not a mistake, it’s a feature!
But other times, it’s a genuine glitch. A failure in the signaling pathways that tell the cell to pinch in two. Imagine a cellular emergency: “Warning! Division initiated! Cytokinesis machinery failing! Repeat: Cytokinesis machinery failing! Abort splitting! Retreat! Retreat!” And the cell, in its infinite wisdom (or lack thereof), just says, “Okay, no splitting it is.”
The consequences of this "no splitting" can be pretty serious. A multinucleated cell can have altered gene expression, making it produce too much of certain proteins and not enough of others. Its metabolism can go haywire. It might become less efficient at its job, or worse, it could become a breeding ground for problems, potentially leading to disease. Imagine your body trying to run with these oversized, unbalanced cells running around. It’s not a good look.

The Molecular Shenanigans: What Goes Wrong?
So, what are the actual molecular players involved in this cellular oopsie? Well, cytokinesis is a complex process. It involves a contractile ring of proteins, primarily actin and myosin (the same stuff that makes your muscles contract, by the way – cool, huh?), that tightens around the middle of the cell, like a drawstring on a bag. This ring constricts, creating a furrow that eventually pinches the cell in two.
For this to happen, a whole cascade of signals needs to fire correctly. There are specific proteins that regulate the formation and contraction of the actin-myosin ring. If any of these proteins are mutated, misexpressed, or simply not activated at the right time, the ring can fail to form, or it can form but not contract properly. It’s like having a beautiful, perfectly threaded needle, but the thread is too weak to sew through the fabric.
One of the key players in regulating this is a group of proteins called Rho GTPases. Think of them as the master switches for the whole operation. If these switches get stuck in the "off" position, or if they’re not receiving the right signals, the contractile ring just won’t get the memo to start squeezing. Oops. And thus, a multinucleated cell is born.
A Case Study in Unintended Consequences (and a Bit of Irony)
Scientists, being the curious bunch they are, have spent a lot of time trying to understand these failures. They use all sorts of clever experiments, often involving genetically modifying cells to disrupt specific genes or pathways. And sometimes, they stumble upon something truly remarkable.

Imagine a researcher trying to study a particular gene involved in cell division. They engineer a cell line where this gene is faulty. Their hypothesis might be that this will cause cells to arrest at a certain stage, or die. But then, they observe something unexpected: the cells continue through mitosis, but they fail to divide. They become these giant, multi-nucleated monstrosities. It's the cellular equivalent of trying to fix a wobbly table leg and accidentally discovering how to create a perfectly spherical planet. Okay, maybe not that dramatic, but you get the idea!
And here’s where the irony kicks in. Sometimes, these intentionally created "failed" cells can actually reveal new insights. By studying why cytokinesis failed in that specific instance, researchers can uncover previously unknown genes or pathways that are essential for normal cell division. It’s like, by breaking something, they accidentally figured out how it’s supposed to work. It’s a bit like my friend Sarah’s cake – a glorious, inedible mess, but maybe it teaches her something about the importance of the final assembly step. Or at least, the importance of keeping squirrels at bay.
When Big Cells Become a Problem: Disease and Development
As I mentioned before, multinucleated cells aren’t always a bad thing. As in muscle cells, they can be part of a specialized function. However, when they arise unintentionally, they can be a sign of trouble. In cancer, for instance, cells are already out of control, dividing haphazardly. If they also start skipping cytokinesis, they can become even more aggressive and resistant to treatment. Imagine a cancer cell that’s not only multiplying rapidly but also becoming a giant, multi-talented mutant. Not ideal for the immune system trying to fight it off.
Multinucleation can also be linked to developmental disorders. Errors in cell division and differentiation are fundamental to how an organism grows. If cells are failing to divide properly early in development, it can lead to significant structural abnormalities. It’s like trying to build a house with faulty bricks – the whole structure is compromised from the start.

The study of these cells, therefore, isn't just academic curiosity. It has real-world implications for understanding and potentially treating diseases. If we can figure out exactly why a cell fails to undergo cytokinesis, we might be able to develop therapies to correct it, or at least prevent it from happening in contexts where it's detrimental.
The Future of "Giant" Cells
So, the next time you think about cells, remember that it's not always a simple case of one becoming two. Sometimes, it's one becoming one, but a much, much bigger one. These multinucleated cells, born from a skipped step in the cell cycle, are a testament to the complexity and sometimes the sheer stubbornness of cellular processes.
Researchers continue to unravel the intricate dance of cytokinesis, identifying the key players and the delicate timing required for a successful cell division. And who knows what else we’ll discover? Maybe we’ll find new ways to harness the power of multinucleation for regenerative medicine, or develop even more targeted cancer therapies. For now, though, let’s just appreciate the fact that most of our cells are pretty good at their jobs, and when they do mess up, they provide us with fascinating puzzles to solve. It’s a messy, wonderful world inside us, isn't it?
And hey, next time you’re baking something complicated, and it doesn't quite turn out as planned, just remember the cells. They have it harder, and sometimes, they end up with a giant, undivided blob too. Just hopefully, yours doesn’t have multiple nuclei. That would be… a bit much.
