A Buffer Contains Significant Amounts Of Ammonia And Ammonium Chloride

Ever wonder how some of your favorite everyday products manage to stay perfectly balanced, neither too acidic nor too basic? It’s like a tiny, invisible superhero is at work behind the scenes! Today, we're diving into the fascinating world of a chemical setup that's both incredibly useful and surprisingly cool: a buffer solution packed with significant amounts of ammonia and ammonium chloride. Forget dusty textbooks; this is chemistry that actually makes a difference in the real world, from the food we eat to the medicines we rely on!
The Magic of Balance: What's a Buffer, Anyway?
So, what exactly is this "buffer" we're talking about? Imagine you're trying to keep a delicate ecosystem in your aquarium perfectly stable. Even a small change in something like CO2 can cause big problems. A buffer solution acts in a very similar way for chemical reactions. Its primary job is to resist changes in pH. Think of pH as the measure of how acidic or basic a solution is – on a scale from 0 (very acidic) to 14 (very basic), with 7 being neutral.
Without a buffer, if you added even a tiny amount of acid or base to a solution, its pH would swing wildly. This is a big no-no for many biological and chemical processes. For instance, your blood needs to stay within a very narrow pH range to keep you healthy. If it gets too acidic or too basic, serious problems arise. That’s where buffers come in, like unsung heroes of chemical stability.
The Dynamic Duo: Ammonia and Ammonium Chloride
Now, let's get to our star players: ammonia (NH₃) and ammonium chloride (NH₄Cl). This particular combination creates what’s known as an ammonia-ammonium buffer. It’s a fantastic example of a weak base buffer system. Why is this combination so special?
Here's the cool part: ammonia is a weak base, and ammonium chloride, when dissolved in water, provides the conjugate acid, the ammonium ion (NH₄⁺). These two components, the weak base and its conjugate acid, are the perfect partners to maintain a stable pH. They are in a constant, dynamic equilibrium. If you try to add an acid to this solution, the ammonia (NH₃) is there to grab onto the extra hydrogen ions (H⁺) from the acid, converting them into ammonium ions (NH₄⁺). This prevents the pH from dropping drastically.
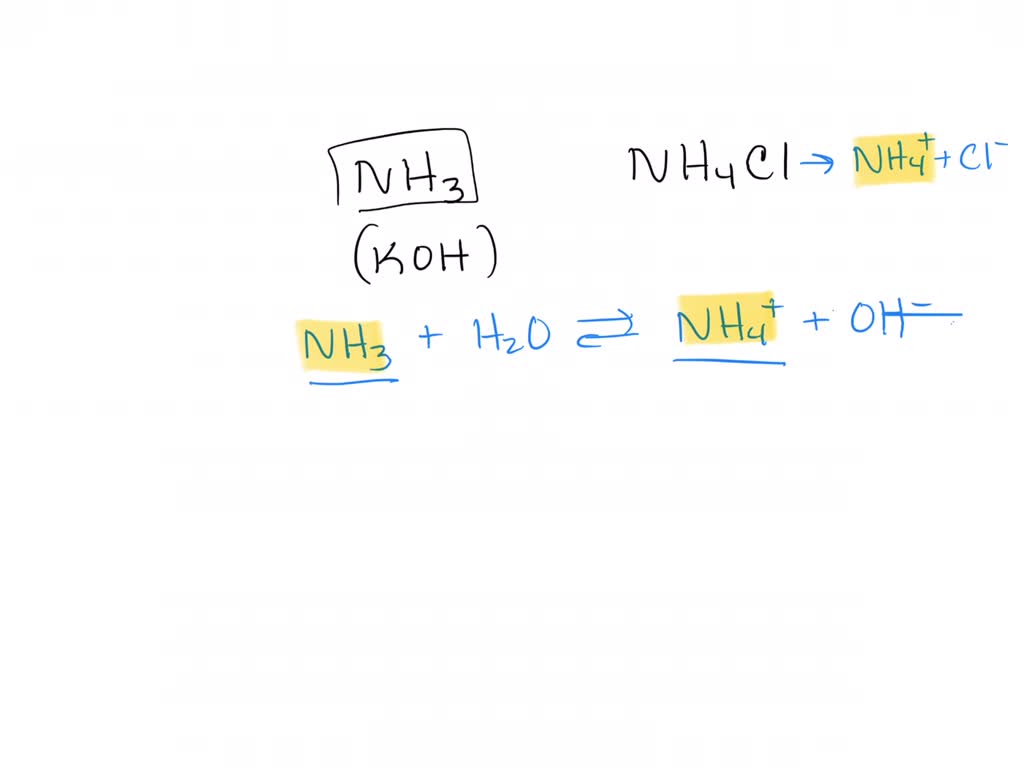
Conversely, if you try to add a base, the ammonium ions (NH₄⁺) are ready to donate their hydrogen ions (H⁺), effectively neutralizing the added base and preventing the pH from rising too much. It’s like having a molecular tug-of-war team that always keeps the rope perfectly centered, no matter who tries to pull harder.
Where Do We See This Buffer in Action?
The applications of an ammonia-ammonium buffer are surprisingly widespread:

- Biochemistry Labs: In countless biological experiments, maintaining a stable pH is crucial for enzymes to function correctly and for cells to survive. Researchers often use this buffer to create the ideal environment for their studies. It’s essential for growing bacterial cultures and performing sensitive protein analysis.
- Medical Applications: While the body has its own sophisticated buffering systems, similar principles are used in medical preparations. Think about certain intravenous solutions or diagnostic tests where precise pH control is paramount.
- Industrial Processes: In various manufacturing settings, from textile dyeing to chemical synthesis, precise pH control is vital for product quality and reaction efficiency. This buffer can play a role in ensuring consistent results.
- Aquaculture: For fish farmers, maintaining the correct pH in fish tanks and ponds is critical for the health and survival of their stock. Ammonia-based buffers can help manage the aquatic environment.
Why It's So Useful (and Maybe Even a Little Fun!)
The beauty of the ammonia-ammonium buffer lies in its effectiveness and its relative affordability. It’s a robust system that can handle moderate additions of acids or bases without significant pH shifts. This reliability makes it a go-to choice for many applications where precision and stability are non-negotiable.
Think of it like this: Imagine you're trying to bake a cake. The recipe calls for precise measurements, and even small deviations can mess up the final product. A buffer is like a smart ingredient in the chemical world that ensures everything stays within the perfect range, allowing other reactions or processes to unfold as intended. It’s the unsung hero that keeps things from going haywire!
So, the next time you hear about a chemical solution being "buffered," remember the clever work of substances like ammonia and ammonium chloride. They’re quietly ensuring that many of the processes we depend on daily, from biological research to industrial production, run smoothly and efficiently. It's a fantastic example of how fundamental chemistry can have a profound and practical impact on our lives!
