A 30.5 Gram Sample Of Glucose Contains Mol Of Glucose

Hey there, science adventurers and sweet tooth enthusiasts! Get ready to dive into a world of tiny, invisible building blocks that make up everything we love, especially our delicious energy sources. Today, we're going to unravel a little mystery that might sound a bit like a secret code, but trust me, it's as fun as finding an extra fry at the bottom of the bag!
So, imagine you've got this super cool, tiny speck of glucose. Not just any glucose, mind you, but a perfectly measured, scientifically precise amount weighing in at a whopping, drumroll please... 30.5 grams! That sounds like a lot, right? Like, maybe enough to sweeten a whole cake? Well, in the grand scheme of things, it's actually a pretty small, manageable chunk. Think of it like a single, perfect sprinkle on a giant ice cream sundae. You know it’s there, it contributes to the deliciousness, but it’s not the whole party.
This 30.5-gram party of glucose is about to get even more interesting. We're going to talk about its moles. Yep, moles! Now, before you start picturing tiny furry creatures scurrying around your sugar bowl, let me assure you, these are not the garden variety kind. These are chemistry moles. And let me tell you, these chemistry moles are the real MVPs of the molecular world.
Think of a mole like a magical counting unit. It’s not a weight, it’s not a volume, it’s a number. A ridiculously, astronomically, mind-bogglingly HUGE number. It’s the kind of number that makes your brain do a little happy dance because it’s so unbelievably big. It’s like trying to count all the stars in the sky, or all the grains of sand on all the beaches, but even bigger! This number, this magical mole number, is approximately 602,200,000,000,000,000,000,000. Say that ten times fast! It’s called Avogadro's Number, and it's the secret handshake of chemists everywhere.
So, what does our little 30.5-gram chunk of glucose have to do with this gigantic mole number? Well, it turns out that our 30.5-gram sample is playing host to a specific amount of these glucose molecules, and when we count them up using our super-powered mole measuring stick, we find out something truly awesome.

This 30.5-gram sample of glucose contains, ta-da! … drumroll again… 0.169 mol of glucose!
There you have it! Our seemingly simple 30.5 grams of sweetness is actually a bustling metropolis of approximately 0.169 moles of glucose molecules. That means there are roughly 101,771,800,000,000,000,000,000 individual glucose molecules chilling in that little sample. Can you even wrap your head around that? It’s more glucose molecules than there are people on Earth, multiplied by, well, a lot!
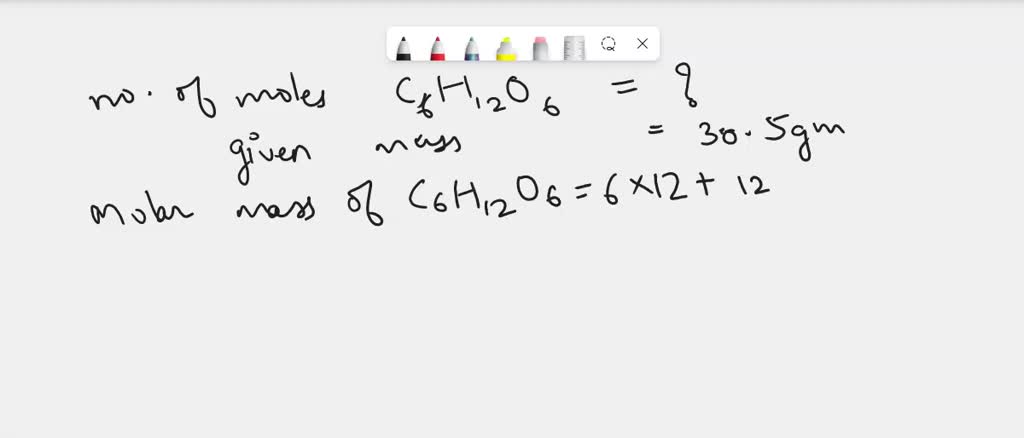
Why is this so cool? Because this is how scientists understand how much of a substance they're working with. It’s like knowing you have enough flour for exactly 5 cakes, or enough sprinkles for a million cupcakes. Knowing the number of moles helps us predict how things will react, how they'll behave, and how we can use them. It’s the secret language of how things are put together!
So, next time you’re enjoying a piece of fruit, a spoonful of honey, or even that little energy boost from a sugary drink, remember the invisible dance happening at the molecular level. Remember that even a tiny amount of something like glucose is actually a colossal gathering of individual molecules, all counted up by our amazing friend, the mole. It’s a sweet, sweet reminder of the incredible complexity and wonder that exists all around us, even in the simplest of things!
Isn't science just the most delicious thing ever? It’s all about finding the extraordinary in the ordinary, and this little glucose sample is proof positive. So go forth, and be amazed by the invisible world of moles!
